
-
 Israeli strike in south Lebanon kills 13 security personnel
Israeli strike in south Lebanon kills 13 security personnel
-
Will The Wise wins Topham as tragedy strikes Gold Dancer

-
 Over 100,000 worshippers perform Friday prayers at Al-Aqsa
Over 100,000 worshippers perform Friday prayers at Al-Aqsa
-
Teen star Seixas claims stage five to close on Basque Tour victory

-
 War's impact on fertilisers stirs food producer fears
War's impact on fertilisers stirs food producer fears
-
US inflation surges to 3.3% as Iran war impact bites

-
 Thais fete new year with family despite fuel price spike
Thais fete new year with family despite fuel price spike
-
Scheffler scrambles, Rose stumbles early at Masters

-
 On Iran truce, all sides want bigger China role, but does China?
On Iran truce, all sides want bigger China role, but does China?
-
Sinner eases into Monte Carlo semi-final against Zverev

-
 Inter skipper Martinez suffers calf injury
Inter skipper Martinez suffers calf injury
-
Ukrainians sceptical as Kremlin orders Easter truce

-
 Arteta urges Arsenal to pile pressure on Man City in title race
Arteta urges Arsenal to pile pressure on Man City in title race
-
Pay fears grow for US security workers in shutdown

-
 Hungary rivals rally crowds in closing strait of election campaign
Hungary rivals rally crowds in closing strait of election campaign
-
Swede goes on trial for pressuring wife to sell sex

-
 US inflation surges 3.3% as Iran war impact bites
US inflation surges 3.3% as Iran war impact bites
-
Vance warns Iran not to 'play' US at talks in Pakistan

-
 Fernandez remains out despite apology: Chelsea boss Rosenior
Fernandez remains out despite apology: Chelsea boss Rosenior
-
Dortmund defender Schlotterbeck extends contract until 2031

-
 De Zerbi vows to save troubled Spurs from relegation
De Zerbi vows to save troubled Spurs from relegation
-
Antwerp port reopens to North Sea shipping after oil spill

-
 Stocks mixed, oil steadies on guarded optimism for Iran ceasefire
Stocks mixed, oil steadies on guarded optimism for Iran ceasefire
-
Sinner eases into Monte Carlo semi-finals

-
 France's Macron talks war, peace and basketball with Pope Leo
France's Macron talks war, peace and basketball with Pope Leo
-
Fernandez apologised over comments about his future: Chelsea's Rosenior

-
 Coach Spalletti signs new Juve deal until 2028
Coach Spalletti signs new Juve deal until 2028
-
AI chatbots offer children harm as if it were help, says activist

-
 'Grumpy' Guardiola wants Silva to stay at Man City for life
'Grumpy' Guardiola wants Silva to stay at Man City for life
-
Zverev beats Fonseca to reach Monte Carlo semi-finals

-
 Scheffler, Rose to chase McIlroy with early Masters starts
Scheffler, Rose to chase McIlroy with early Masters starts
-
Celine Dion's Paris concerts promise to spin the money on and on

-
 Stocks climb, oil steadies on guarded optimism over Iran war ceasefire
Stocks climb, oil steadies on guarded optimism over Iran war ceasefire
-
Irish govt to meet farmers, hauliers over fuel cost fears

-
 Injured Bayern starlet Karl to miss Real return leg
Injured Bayern starlet Karl to miss Real return leg
-
US-Iran talks in Pakistan uncertain as sides trade accusations

-
 Oil spill snarls shipping traffic in Antwerp port
Oil spill snarls shipping traffic in Antwerp port
-
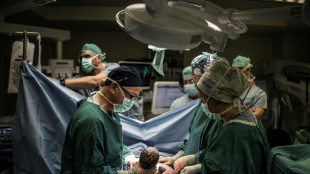
Giving birth in a shelter in Israel
-
 Five things to know about the planned Iran-US talks in Islamabad
Five things to know about the planned Iran-US talks in Islamabad
-
Slot feels 'complete support' from Liverpool chiefs despite slump

-
 Kyiv books tentative diplomatic coup with Iran war forays
Kyiv books tentative diplomatic coup with Iran war forays
-
Teenager shines as Britain seize control of BJK Cup tie with Australia

-
 Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader
Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader
-
Sleepy seal diverts traffic in Australian seaside town

-
 Artemis astronauts to shed light on space health risks
Artemis astronauts to shed light on space health risks
-
Pakistan prepares to host US-Iran talks, as Lebanon fighting continues

-
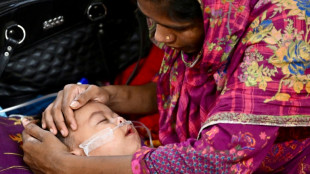 Vaccine gaps fuel Bangladesh's deadly measles crisis
Vaccine gaps fuel Bangladesh's deadly measles crisis
-
Fish furore fuels fierce election in India's West Bengal

-
 Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G
Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G
-
Myanmar junta chief sworn in as president


BioLargo Subsidiary Clyra Medical Technologies Secures First Stocking Order with Advanced Solution for ViaCLYR(TM) Wound Irrigation Solution
Initial Distribution Launch Marks Transition to Revenue-Generating Operations
WESTMINSTER, CALIFORNIA / ACCESS Newswire / February 9, 2026 / BioLargo, Inc. (OTCQX:BLGO), a cleantech and life sciences innovator, announced today that its subsidiary, Clyra Medical Technologies, Inc., has received its first commercial stocking order from Advanced Solution, LLC for ViaCLYR™, Clyra's FDA-cleared wound irrigation solution. The order marks the start of commercial distribution and represents Clyra's transition from development-stage operations to early revenue generation.
The stocking order initiates product availability through Advanced Solution's national distribution platform, supporting sales to hospitals, clinics, and healthcare providers across the United States. BioLargo views this milestone as the first in a series of planned commercial and revenue-related announcements as Clyra advances its market rollout.
ViaCLYR™ is a highly effective, tissue safe, long-acting wound irrigation solution that can be used for acute and chronic wounds and burns. This unique, clear, odorless, non-irritating solution is 510(k) cleared by the FDA and indicated for acute and chronic wounds. It has an extremely high antimicrobial activity as a preservative in solution with over 99.9999% kill rate (up to 7 log reduction) with sustained efficacy up to 72 hours.
"This initial commercial order reflects years of disciplined development, regulatory execution, and commercial preparation," said Steve Harrison, Clyra's Chief Executive Officer. "With ViaCLYR™ now entering the distribution channel, we are beginning the transition from innovation to execution, laying the groundwork for scalable revenue growth as adoption expands."
Dennis P. Calvert, Chairman of Clyra Medical Technologies and President and CEO of BioLargo, Inc., added, "This milestone represents a tangible step forward in BioLargo's strategy of developing differentiated technologies and advancing them into commercial markets through strong partnerships. We believe Clyra is well positioned to build momentum as product availability expands and market awareness grows."
Clyra's commercial readiness reflects the completion of key foundational elements, including FDA 510(k) clearance, implementation of ISO 13485-certified quality systems, validated manufacturing processes, and clinical evaluations supporting real-world use. As distribution begins, Clyra plans to advance additional commercialization initiatives and expand its product portfolio over time.
About Clyra Medical Technologies
Clyra Medical Technologies, Inc., a partially-owned subsidiary of BioLargo, Inc., focuses on infection control and advanced wound care. Founded in 2012, the company develops and commercializes wound care solutions based on its proprietary Copper-Iodine Complex Technology. Clyra's product portfolio features FDA-cleared medical devices that leverage this patented technology to deliver superior wound care outcomes. The company continues to advance its research and development efforts, exploring expanded indications for its unique formulations and their synergistic applications with complementary wound care dressings and mechanical devices. Through strategic partnerships with leading distribution and marketing organizations, Clyra ensures its innovative products reach healthcare providers and patients who need them most.
About Advanced Solution
Advanced Solution LLC (https://advancedsolution.health) specializes in advanced wound care biologics, regenerative technologies, and medical devices. As an FDA-registered Tissue Dispensary Intermediary (TDI), Advanced Solution partners with healthcare providers, hospitals, and clinics across the United States to deliver innovative, outcomes-driven products that improve patient care and operational efficiency. Headquartered in Carlisle, Pennsylvania, Advanced Solution represents a portfolio of market-leading brands and collaborates closely with manufacturers, clinicians, and healthcare organizations to bring cutting-edge therapies to market. With a focus on compliance, education, and strategic commercialization, the company continues to set the standard for excellence in distribution and partnership within the wound care and life sciences industries.
About BioLargo, Inc.
BioLargo, Inc. (OTCQX:BLGO) is a cleantech and life sciences innovator and engineering services solution provider. Our core products address PFAS contamination, achieve advanced water and wastewater treatment, control odor and VOCs, improve air quality, enable energy-efficiency and safe on-site energy storage, and control infections and infectious disease. Our approach is to invent or acquire novel technologies, develop them into product offerings, and extend their commercial reach through licensing and channel partnerships to maximize their impact. See our website at www.BioLargo.com.
Contact Information
Dennis P. Calvert
President and CEO, BioLargo, Inc.
888-400-2863
Safe Harbor Act
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These forward-looking statements include without limitation those about BioLargo's (the "Company") expectations regarding anticipated revenue; and plans for future operations. These statements involve risks and uncertainties, and actual results may differ materially from any future results expressed or implied by the forward-looking statements. Risks and uncertainties include without limitation: the effect of regional economic conditions on the Company's business, including effects on purchasing decisions by consumers and businesses; the ability of the Company to compete in markets that are highly competitive and subject to rapid technological change; the ability of the Company to manage frequent introductions and transitions of products and services, including delivering to the marketplace, and stimulating customer demand for, new products, services, and technological innovations on a timely basis; the dependency of the Company on the performance of distributors of the Company's products. More information on these risks and other potential factors that could affect the Company's business and financial results is included in the Company's filings with the SEC, including in the "Risk Factors" and "Management's Discussion and Analysis of Financial Condition and Results of Operations" sections of the Company's most recently filed periodic reports on Form 10-K and Form 10-Q and subsequent filings. The Company assumes no obligation to update any forward-looking statements or information, which speak as of their respective dates.
SOURCE: BioLargo, Inc.
View the original press release on ACCESS Newswire
Ch.P.Lewis--AT




