
-
 Airbus profits slide as deliveries drop
Airbus profits slide as deliveries drop
-
Trump hails British 'friends' as king visits

-
 Hungary's PM-elect Magyar offers to meet Ukraine's Zelensky in June
Hungary's PM-elect Magyar offers to meet Ukraine's Zelensky in June
-
Man pleads guilty to plotting attack on Taylor Swift concert

-
 New pirate group behind latest Somali hijacking: officials
New pirate group behind latest Somali hijacking: officials
-
Swiss court dismisses corruption case against late Uzbek leader's daughter

-
 Frenchman Godon wins Romandie prologue, Pogacar fifth
Frenchman Godon wins Romandie prologue, Pogacar fifth
-
Trump hails British as 'friends' as king visits amid Iran tensions

-
 Will fuel shortages ruin summer vacations?
Will fuel shortages ruin summer vacations?
-
Peace efforts stall as US examines latest Iran proposal

-
 Mali faces advancing rebels in 'difficult' situation
Mali faces advancing rebels in 'difficult' situation
-
Monk ends barefoot Sri Lanka trek with a dog and plea for peace

-
 Macron urges Andorra to 'move forwards' on decriminalising abortion
Macron urges Andorra to 'move forwards' on decriminalising abortion
-
German bid to rescue 'Timmy' the whale passes key hurdle

-
 US Fed expected to keep rates steady as Iran war effects ripple
US Fed expected to keep rates steady as Iran war effects ripple
-
UAE pulls out of OPEC oil cartels citing 'national interests'

-
 Crude back above $110 on Strait stalemate fears
Crude back above $110 on Strait stalemate fears
-
Banking giant JP Morgan becomes Olympics sponsor

-
 Emotional Stones announces Man City exit after golden decade
Emotional Stones announces Man City exit after golden decade
-
Jazz legend John Coltrane's son hits the high notes

-
 John Stones to leave Manchester City after 10 years
John Stones to leave Manchester City after 10 years
-
Croatia, Bosnia sign major gas pipeline deal

-
 Champions League semi-final like a first date: Atletico's Koke
Champions League semi-final like a first date: Atletico's Koke
-
Sinner queries schedule, surges into Madrid Open quarters

-
 ICC orders $8.5mn compensation for victims of Malian war criminal
ICC orders $8.5mn compensation for victims of Malian war criminal
-
EU parliament adopts new rules to protect cats, dogs

-
 EU lawmakers back blockbuster long-term budget
EU lawmakers back blockbuster long-term budget
-
German rescuers launch new bid to free stranded whale

-
 Man pleads guilty in Austria to plotting attack on Taylor Swift concert
Man pleads guilty in Austria to plotting attack on Taylor Swift concert
-
Climbers open Everest route past dangerous ice block

-
 Indian billionaire's son offers home for Escobar's hippos
Indian billionaire's son offers home for Escobar's hippos
-
Iranian Vafaei capable of great things, says beaten rival Trump

-
 Comedian Kimmel hits back at criticism over Melania Trump joke
Comedian Kimmel hits back at criticism over Melania Trump joke
-
Man goes on trial in Austria over Taylor Swift concert attack plan

-
 South Korean court increases ex-first lady's graft sentence
South Korean court increases ex-first lady's graft sentence
-
Bullying claims 'nonsense', actress Rebel Wilson tells Sydney court

-
 BP reports huge profit rise in first quarter
BP reports huge profit rise in first quarter
-
Crude extends gains, stocks drop as Trump considers latest Iran proposal

-
 How China block of AI deal could stop 'Singapore-washing'
How China block of AI deal could stop 'Singapore-washing'
-
North Korean executions rose dramatically during Covid: report

-
 Budget airlines first to cut flights as jet fuel prices soar
Budget airlines first to cut flights as jet fuel prices soar
-
Simeone, Atletico chasing redemption against Arsenal

-
 'Bring it on', says Rice as Arsenal chase Champions League history
'Bring it on', says Rice as Arsenal chase Champions League history
-
US says examining latest Iran proposal

-
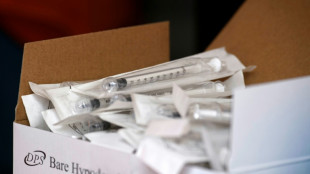 S. Korea probes syringe hoarding as war hits plastic makers
S. Korea probes syringe hoarding as war hits plastic makers
-
Australia aims to tax tech giants unless they pay news outlets

-
 Bangladesh's tigers stalk uncertain future in Sundarbans
Bangladesh's tigers stalk uncertain future in Sundarbans
-
Horses unlikely saviours for those who serve in uniform

-
 Crude extends gains as Trump considers latest Iran proposal
Crude extends gains as Trump considers latest Iran proposal
-
Nations to kick off world-first fossil fuel exit talks


IGC Pharma Advances Caregiver Engagement as Phase 2 CALMA Trial Advances Final Enrollment Phase
Outreach initiative supports awareness of agitation and patient engagement as enrollment approaches completion
POTOMAC, MD / ACCESS Newswire / April 28, 2026 / IGC Pharma, Inc. (NYSE American:IGC) ("IGC" or the "Company"), a clinical-stage biotechnology company developing therapeutics for Alzheimer's disease, today announced its participation in the caregiver-focused podcast Autumn Is Here, as part of its broader effort to increase awareness of agitation associated with Alzheimer's disease and support patient engagement in its ongoing Phase 2 CALMA clinical trial, which has recently reached approximately 80% patient enrollment and is advancing toward completion.

The episode featured Margarita Venegas, Psychometric Training Manager, and Barbara Forero, Clinical Neuropsychologist & Recruitment Coordinator at IGC Pharma, in a discussion focused on recognizing agitation as a distinct and clinically significant symptom of Alzheimer's disease. The conversation was hosted by Francine Crawford, creator of Autumn Is Here, who brings a personal caregiving perspective.
Supporting Clinical Progress Through Awareness
As the CALMA trial advances toward its final phase of enrollment, increasing awareness of agitation remains an important component of both patient care and clinical trial participation. The Company believes that caregiver education can support earlier recognition of symptoms and improve engagement in clinical studies evaluating potential treatment options.
During the episode, participants discussed how agitation may present in patients, how it differs from other behavioral symptoms, and why it is often under-recognized despite its significant impact on patients and caregivers.
Connecting Research and the Caregiver Community
"Engaging with caregivers is an important part of advancing Alzheimer's research," said Ram Mukunda, Chief Executive Officer of IGC Pharma. "As CALMA progresses toward enrollment completion, outreach and education play a role in supporting awareness of agitation and the importance of continued research in this area."
The Company continues to prioritize outreach efforts that connect clinical research with patients, caregivers, and healthcare professionals as it advances the CALMA clinical trial toward completion.
The full podcast episode is available here: Link.
About IGC Pharma (dba IGC):
IGC Pharma (NYSE American:IGC) is a clinical-stage biotechnology company leveraging AI to develop innovative treatments for Alzheimer's and metabolic disorders. Our lead asset, IGC-AD1, is a therapy currently in a Phase 2 trial (CALMA) for agitation in Alzheimer's dementia. Our pipeline includes TGR-63, targeting amyloid plaques, and early-stage programs focused on neurodegeneration, tau proteins, and metabolic dysfunctions. We integrate AI to accelerate drug discovery, optimize clinical trials, and enhance patient targeting. With a complete patent portfolio and a commitment to innovation, IGC Pharma is advancing breakthrough therapies.
Forward-Looking Statements:
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements are based on current expectations and involve risks and uncertainties that could cause actual results to differ materially, including risks related to the Company's ability to complete enrollment in its Phase 2 CALMA trial within anticipated timeframes, demonstrate safety and efficacy, the timing of data readouts, regulatory approvals, and other factors discussed in the Company's filings with the U.S. Securities and Exchange Commission (SEC), including its most recent Annual Report on Form 10-KT. The Company undertakes no obligation to update these statements.
Contact Information:
Walter Frank / John Nesbett
IMS Investor Relations
[email protected]
(203) 972-9200
SOURCE: IGC Pharma, Inc.
View the original press release on ACCESS Newswire
B.Torres--AT



