
-
 Six new caps for France for women's Six Nations opener
Six new caps for France for women's Six Nations opener
-
Calls for US-Iran truce to extend to Lebanon after Israeli strikes

-
 Nepal ex-PM Oli gives defiant message after release from custody
Nepal ex-PM Oli gives defiant message after release from custody
-
Despite Middle East truce, airlines fear long-term disruptions

-
 Memorial: Russia's Nobel Prize winning rights group facing 'extremism' ban
Memorial: Russia's Nobel Prize winning rights group facing 'extremism' ban
-
Artemis crew's families enthralled by messages from space

-
 Champions Cup 'heartbreak' driving Toulouse revenge mission
Champions Cup 'heartbreak' driving Toulouse revenge mission
-
Shallow Indonesian quake damages houses, injures residents

-
 Nepal ex-PM Oli released from custody after 12 days: police
Nepal ex-PM Oli released from custody after 12 days: police
-
'Chills': Artemis astronauts say lunar flyby still washing over them

-
 Ukraine lets firms deploy air defences against Russian attacks
Ukraine lets firms deploy air defences against Russian attacks
-
Mountain-made: Balkan sheepdog eyes future beyond the hills

-
 Escaped wolf forces school closure in South Korea
Escaped wolf forces school closure in South Korea
-
Three ways Orban gives himself an edge in Hungary's vote

-
 Trump says US military to stay deployed near Iran until 'real agreement' reached
Trump says US military to stay deployed near Iran until 'real agreement' reached
-
Gender-row boxer Lin targets Asian Games after bronze on comeback

-
 US-Iran truce shows cracks as war flares in Lebanon
US-Iran truce shows cracks as war flares in Lebanon
-
In Romania, many Hungarians root for Orban in vote

-
 Home where young Bowie dreamt of 'fame' to open to public
Home where young Bowie dreamt of 'fame' to open to public
-
Crude rises, stocks fall on fears over nascent Iran ceasefire

-
 Waiting for DeepSeek: new model to test China's AI ambitions
Waiting for DeepSeek: new model to test China's AI ambitions
-
You're being watched: Japan battles online abuse of athletes

-
 US court expedites Anthropic's legal battle with Department of War
US court expedites Anthropic's legal battle with Department of War
-
Badminton to trial synthetic shuttlecocks because of feather shortage

-
 Firm, fast Augusta set to test golf's best in 90th Masters
Firm, fast Augusta set to test golf's best in 90th Masters
-
BTS to kick off world tour after landmark Seoul comeback

-
 Grand National had to change to survive, says former winning jockey
Grand National had to change to survive, says former winning jockey
-
Maple syrup or nutella? PM Carney calls Canadian Artemis astronaut

-
 Comedy duo Flight of the Conchords reunion gigs sell out in minutes
Comedy duo Flight of the Conchords reunion gigs sell out in minutes
-
US-Iran truce enters second day as war flares in Lebanon

-
 Trump blasts NATO after closed-door Rutte meeting
Trump blasts NATO after closed-door Rutte meeting
-
Houston, we have a problem ... with the toilet

-
 FireFox Gold Expands the East Zone to the Southwest with Ongoing Grid Drilling at the Mustajärvi Gold Project, Finland
FireFox Gold Expands the East Zone to the Southwest with Ongoing Grid Drilling at the Mustajärvi Gold Project, Finland
-
Bolt Metals Corp. Announces Appointment of Chief Financial Officer and Corporate Secretary

-
 Slot admits Liverpool in 'survival mode' in PSG defeat
Slot admits Liverpool in 'survival mode' in PSG defeat
-
Trump makes up with Sahel juntas, with eye on US interests

-
 Tiger Woods drug records to be subpoenaed by prosecutors
Tiger Woods drug records to be subpoenaed by prosecutors
-
England's Rai wins Par-3 Contest to risk Masters curse

-
 Brazil's Chief Raoni backs Lula in elections
Brazil's Chief Raoni backs Lula in elections
-
Trump to discuss leaving NATO in meeting with Rutte

-
 Atletico punish 10-man Barcelona, take control of Champions League tie
Atletico punish 10-man Barcelona, take control of Champions League tie
-
Dominant PSG leave Liverpool right up against it in Champions League tie

-
 Meta releases first new AI model since shaking up team
Meta releases first new AI model since shaking up team
-
Tehran residents relieved but divided by Trump truce

-
 Vance says up to Iran if it wants truce to 'fall apart' over Lebanon
Vance says up to Iran if it wants truce to 'fall apart' over Lebanon
-
US, Iran truce hangs in balance as war flares in Lebanon

-
 Scale of killing in Lebanon 'horrific': UN rights chief
Scale of killing in Lebanon 'horrific': UN rights chief
-
'Ketamine Queen' jailed for 15 years over Matthew Perry drugs

-
 Betis earn draw in Europa League quarter-final at Braga
Betis earn draw in Europa League quarter-final at Braga
-
Buttler hits form with IPL fifty as Gujarat win last-ball thriller


ClearPoint Neuro Announces Development and Demonstration of the Company's Proprietary Robotic Neuro-Navigation System
New Product Category Will Enable Added Flexibility to Support Commercial Launches of Cell and Gene Therapy
SOLANA BEACH, CALIFORNIA / ACCESS Newswire / October 1, 2025 / ClearPoint Neuro, Inc. (Nasdaq:CLPT) (the "Company"), a global device, cell, and gene therapy-enabling company offering precise navigation to the brain and spine, today formally announced the development and demonstration of the Company's proprietary Robotic Neuro-Navigation System. This new product category will enable the ClearPoint Neuro Navigation software to operate the KUKA LBR Med Robotic Arm to support all minimally invasive cranial surgical procedures including cell and gene therapy infusions, laser catheter placement, biopsy workflows and deep brain stimulation and stereotactic EEG lead placements. The Company will demonstrate a prototype of this robotic system at the 75th Annual Congress of Neurological Surgeons (CNS) in Los Angeles October 13th-15th.
"Everything we do at ClearPoint Neuro is viewed through the lens of 'how does this help us extend our lead in neuro drug delivery and help our partners prepare for the commercialization of these new cell and gene therapies," commented Jeremy Stigall, CBO and GM of the Biologics and Drug Delivery Business at ClearPoint. "Many of our BioPharma partners have the same dilemma that ClearPoint is helping to solve. On one side, they see the clear value of MRI guidance for drug delivery clinical trials and regulatory submissions, yet the requirements for MRI guidance can be viewed as narrow in flexibility and surgeon choice and can, therefore, limit adoption. On the other side, BioPharma understands that providing too much flexibility with the surgical technique can lead to less-than-optimal results because the ability to train on the crucial surgical technique in a consistent and organized manner can be lost with that flexibility. Just like a lean manufacturing line, you cannot increase quality and reduce procedure time and cost without consistency and repetition. BioPharma needs a single navigation partner that can provide that sweet spot of offering BOTH consistency and flexibility."
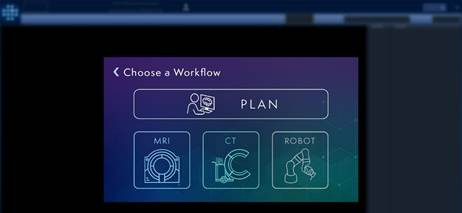
"The addition of the ClearPoint Neuro Robotic Neuro-Navigation System to our portfolio is designed to do exactly that - to provide consistency and flexibility to the commercial launches of various cell and gene therapies," continued Stigall. "First, for consistency, there will be one ClearPoint software planning module which is already used today. This software will be the first step for the surgical team to plan every patient's drug infusion plan, often days before the surgery itself. Second, the surgical team will then decide through which hardware mechanism to execute that surgical plan across three different ClearPoint options: the traditional MRI guided technique, the new ClearPoint 3.0 iCT guided technique in the operating room, and the future ClearPoint Robotic assisted technique also using iCT. Each of these options benefit from the same ClearPoint software workflow, and many advanced drug delivery features. We believe this strategy is unique, and that ClearPoint is best suited to take on this responsibility with our drug delivery portfolio, including co-labeled cannula-based routes-of-administration, pre-clinical CRO testing and live specialist procedural support."
"We have reached a point in medicine that if you close your eyes and imagine the future of neurosurgery, there is no way that robotics are not a significant part of that future," commented Joe Burnett, President and CEO of ClearPoint Neuro. "We have been very thoughtful and deliberate in our approach to enter the neuro robotics market. In years past, a company would have to develop a complete robotic system from scratch and bear the entire development risk and significant costs along the way. Today, by working with KUKA, we can leverage their decades of experience which has produced an FDA cleared and CE marked robotic arm and combine that with the decades of experience we have at ClearPoint, creating a state-of-the-art neuro navigation software to drive the robot in the same 3D space. This dramatically decreases the development cost and accelerates our speed to market. The timing is crucial as we are helping our BioPharma partners to identify regional treatment centers and to add surgical capacity in preparation of more significant commercial case volume in the next couple of years."
The ClearPoint Neuro Robotic Neuro-Navigation System has not yet been submitted to any regulatory body. The Company will continue product development with the goal of commercialization in concert with the approval of multiple neuro cell and gene therapies which are currently under FDA expedited review.
About ClearPoint Neuro
ClearPoint Neuro is a device, cell, and gene therapy-enabling company offering precise navigation to the brain and spine. The Company uniquely provides both established clinical products as well as preclinical development services for controlled drug and device delivery. The Company's flagship product, the ClearPoint Neuro Navigation System, has FDA clearance and is CE-marked. ClearPoint Neuro is engaged with healthcare and research centers in North America, Europe, Asia, and South America. The Company is also partnered with the most innovative pharmaceutical/biotech companies, academic centers, and contract research organizations, providing solutions for direct central nervous system delivery of therapeutics in preclinical studies and clinical trials worldwide. To date, thousands of procedures have been performed and supported by the Company's field-based clinical specialist team, which offers support and services to our customers and partners worldwide. For more information, please visit www.clearpointneuro.com.
Forward-Looking Statements
This press release contains forward-looking statements within the context of the federal securities laws, including the Company's product development plans, and the expectation for commercial objectives, potential benefits, and timing of a product that remains in the initial stage of development. These forward-looking statements are based on management's current expectations and are subject to the risks inherent in the business, which may cause the Company's actual results to differ materially from those expressed in or implied by forward-looking statements. Particular uncertainties and risks include those relating to: macroeconomic and inflationary conditions; regulatory and policy uncertainty; the introduction of or changes in tariffs, sanctions, or trade barriers; changes in monetary policy; geopolitical trends, such as protectionism and economic nationalism; future revenue from sales of the Company's products and services; the Company's ability to market, commercialize and achieve broader market acceptance for new products and services offered by the Company; the ability of our biologics and drug delivery partners to achieve commercial success, including their use of the Company's products and services in their delivery of therapies; the Company's ability to maintain its current relationships with biologics and drug delivery partners or enter into new relationships with such partners; the Company's expectations, projections and estimates regarding expenses, future revenue, capital requirements, and the availability of and the need for additional financing; the Company's ability to obtain additional funding to support its research and development programs; the ability of the Company to manage the growth of its business; the Company's ability to attract and retain its key employees; and risks inherent in the research, development, and regulatory approval of the Company's new products, including the ability to obtain and maintain regulatory clearances; manufacturing scale-up; supply chain and quality system readiness; intellectual property protection and freedom to operate; market acceptance and competitive dynamics. More detailed information on these and additional factors that could affect the Company's actual results are described in the "Risk Factors" section of the Company's Annual Report on Form 10-K for the year ended December 31, 2024, and the Company's Quarterly Report on Form 10-Q for the three months ended June 30, 2025, both of which have been filed with the Securities and Exchange Commission. The Company does not assume any obligation to update these forward-looking statements.
Contact:
Investor Relations:
Danilo D'Alessandro, Chief Financial Officer
(888) 287-9109 ext. 3
[email protected]
SOURCE: ClearPoint Neuro, Inc.
View the original press release on ACCESS Newswire
Y.Baker--AT
