
-
 Trump says Iran has 'no cards' beyond Hormuz control
Trump says Iran has 'no cards' beyond Hormuz control
-
Israeli strike in south Lebanon kills 13 security personnel

-
 Will The Wise wins Topham as tragedy strikes Gold Dancer
Will The Wise wins Topham as tragedy strikes Gold Dancer
-
Over 100,000 worshippers perform Friday prayers at Al-Aqsa

-
 Teen star Seixas claims stage five to close on Basque Tour victory
Teen star Seixas claims stage five to close on Basque Tour victory
-
War's impact on fertilisers stirs food producer fears

-
 US inflation surges to 3.3% as Iran war impact bites
US inflation surges to 3.3% as Iran war impact bites
-
Thais fete new year with family despite fuel price spike

-
 Scheffler scrambles, Rose stumbles early at Masters
Scheffler scrambles, Rose stumbles early at Masters
-
On Iran truce, all sides want bigger China role, but does China?

-
 Sinner eases into Monte Carlo semi-final against Zverev
Sinner eases into Monte Carlo semi-final against Zverev
-
Inter skipper Martinez suffers calf injury

-
 Ukrainians sceptical as Kremlin orders Easter truce
Ukrainians sceptical as Kremlin orders Easter truce
-
Arteta urges Arsenal to pile pressure on Man City in title race

-
 Pay fears grow for US security workers in shutdown
Pay fears grow for US security workers in shutdown
-
Hungary rivals rally crowds in closing strait of election campaign

-
 Swede goes on trial for pressuring wife to sell sex
Swede goes on trial for pressuring wife to sell sex
-
US inflation surges 3.3% as Iran war impact bites

-
 Vance warns Iran not to 'play' US at talks in Pakistan
Vance warns Iran not to 'play' US at talks in Pakistan
-
Fernandez remains out despite apology: Chelsea boss Rosenior

-
 Dortmund defender Schlotterbeck extends contract until 2031
Dortmund defender Schlotterbeck extends contract until 2031
-
De Zerbi vows to save troubled Spurs from relegation

-
 Antwerp port reopens to North Sea shipping after oil spill
Antwerp port reopens to North Sea shipping after oil spill
-
Stocks mixed, oil steadies on guarded optimism for Iran ceasefire

-
 Sinner eases into Monte Carlo semi-finals
Sinner eases into Monte Carlo semi-finals
-
France's Macron talks war, peace and basketball with Pope Leo

-
 Fernandez apologised over comments about his future: Chelsea's Rosenior
Fernandez apologised over comments about his future: Chelsea's Rosenior
-
Coach Spalletti signs new Juve deal until 2028

-
 AI chatbots offer children harm as if it were help, says activist
AI chatbots offer children harm as if it were help, says activist
-
'Grumpy' Guardiola wants Silva to stay at Man City for life

-
 Zverev beats Fonseca to reach Monte Carlo semi-finals
Zverev beats Fonseca to reach Monte Carlo semi-finals
-
Scheffler, Rose to chase McIlroy with early Masters starts

-
 Celine Dion's Paris concerts promise to spin the money on and on
Celine Dion's Paris concerts promise to spin the money on and on
-
Stocks climb, oil steadies on guarded optimism over Iran war ceasefire

-
 Irish govt to meet farmers, hauliers over fuel cost fears
Irish govt to meet farmers, hauliers over fuel cost fears
-
Injured Bayern starlet Karl to miss Real return leg

-
 US-Iran talks in Pakistan uncertain as sides trade accusations
US-Iran talks in Pakistan uncertain as sides trade accusations
-
Oil spill snarls shipping traffic in Antwerp port

-
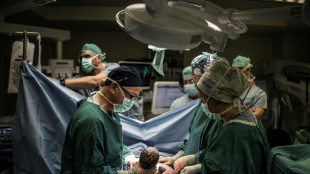 Giving birth in a shelter in Israel
Giving birth in a shelter in Israel
-
Five things to know about the planned Iran-US talks in Islamabad

-
 Slot feels 'complete support' from Liverpool chiefs despite slump
Slot feels 'complete support' from Liverpool chiefs despite slump
-
Kyiv books tentative diplomatic coup with Iran war forays

-
 Teenager shines as Britain seize control of BJK Cup tie with Australia
Teenager shines as Britain seize control of BJK Cup tie with Australia
-
Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader

-
 Sleepy seal diverts traffic in Australian seaside town
Sleepy seal diverts traffic in Australian seaside town
-
Artemis astronauts to shed light on space health risks

-
 Pakistan prepares to host US-Iran talks, as Lebanon fighting continues
Pakistan prepares to host US-Iran talks, as Lebanon fighting continues
-
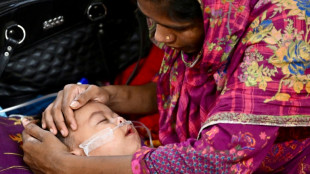
Vaccine gaps fuel Bangladesh's deadly measles crisis
-
 Fish furore fuels fierce election in India's West Bengal
Fish furore fuels fierce election in India's West Bengal
-
Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G


Dogwood Therapeutics Files New Synthetic Halneuron(R) Intellectual Property Protection Projected to Extend Exclusivity Period Up to 2045
- New Composition of Matter Intellectual Property ("IP") Filing Centered on First-in-Class, Fully Synthetically Manufactured Halneuron®-
- New Synthetic Process to be Used for Phase 3 Development Provides Higher Manufacturing Yields and Reduces Costs Versus Naturally Harvested Halneuron® -
ATLANTA, GA / ACCESS Newswire / December 2, 2025 / Dogwood Therapeutics, Inc. (Nasdaq:DWTX) (the "Company"), a development-stage biotechnology company developing new medicines to treat pain and neuropathy, today announced it has filed for new intellectual property protection for synthetic Halneuron®. If granted, the IP's exclusivity period will extend to 2045, before additional patent term restoration extensions.
"We plan to use synthetically manufactured Halneuron® in both Phase 3 development and commercialization," said Greg Duncan, Dogwood Therapeutics Chief Executive Officer. "This new synthetic process builds on our proprietary and best-in-class manufacturing know-how, with potential to reset our exclusivity period for 20 years, thus providing ample opportunity to deliver Halneuron's® full commercial potential."
About Halneuron®
Our lead product candidate, Halneuron®, is in Phase 2b development as a non-opioid, NaV 1.7 inhibitor to treat pain conditions including the neuropathic pain associated with chemotherapy treatment ("CINP"). Halneuron® has been granted fast track designation from the Food and Drug Administration ("FDA") for the treatment of CINP. Success in this Phase 2b study will serve as the basis for the Company engaging the FDA to align on the Halneuron® Phase 3 development program requirements.
About Dogwood Therapeutics
Dogwood Therapeutics (Nasdaq:DWTX) is a development-stage biopharmaceutical company focused on developing new medicines to treat pain and neuropathic disorders. The Dogwood research pipeline includes two first-in-class development candidates, Halneuron® and SP16 IV.
Our lead product candidate, Halneuron®, is in Phase 2b development to treat pain conditions including the neuropathic pain associated with chemotherapy treatment. Halneuron® has been granted fast track designation from the FDA for the treatment of CINP. Halneuron® is a non-opioid, NaV 1.7 analgesic which is a highly specific voltage-gated sodium channel modulator, a mechanism known to be effective for reducing pain transmission. In clinical studies, Halneuron® treatment has demonstrated pain reduction in pain related to general cancer and in pain related to chronic chemotherapy-induced neuropathic pain ("CINP").
SP16 IV is a low-density lipoprotein receptor related protein-1 (LRP1) agonist with potential to treat neuropathy and prevent or repair nerve damage following chemotherapy. SP16 acts as an LRP1 agonist that in turn provides alpha-1-antitrypsin-like activity. Consistent with alpha-1-antitrypsin anti-inflammatory and immunomodulatory actions, SP16 preclinically demonstrated anti-inflammatory (analgesic) action via potential reductions in IL-6, IL-8, IL1B and TNF-alpha levels, as well as potential to repair damaged tissue via increases in pAKT and pERK that regulate fundamental processes like growth, proliferation and survival. The forthcoming SP16 IV Phase 1b CINP trial is fully funded by the National Cancer Institute.
For more information, please visit www.dwtx.com.
Forward-Looking Statements:
Statements in this press release contain "forward-looking statements," within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, that are subject to substantial risks and uncertainties. All statements, other than statements of historical fact, contained in this press release are forward-looking statements. Forward-looking statements contained in this press release may be identified by the use of words such as "anticipate," "believe," "contemplate," "could," "estimate," "expect," "intend," "seek," "may," "might," "plan," "potential," "predict," "project," "suggest," "target," "aim," "should," "will," "would," or the negative of these words or other similar expressions, although not all forward-looking statements contain these words. Forward-looking statements are based on Dogwood's current expectations and are subject to inherent uncertainties, risks and assumptions that are difficult to predict, including risks related to the completion, timing and results of current and future clinical studies relating to Dogwood's product candidates. Further, certain forward-looking statements are based on assumptions as to future events that may not prove to be accurate. These and other risks and uncertainties are described more fully in the section titled "Risk Factors" in the Annual Report on Form 10-K for the year ended December 31, 2024, which has been filed with the Securities and Exchange Commission. Forward-looking statements contained in this announcement are made as of this date, and Dogwood undertakes no duty to update such information except as required under applicable law.
Investor Relations:
CORE IR
(516) 222-2560
[email protected]
SOURCE: Dogwood Therapeutics, Inc.
View the original press release on ACCESS Newswire
Ch.P.Lewis--AT

