
-
 Trump warns Iran better 'get smart soon' and accept nuclear deal
Trump warns Iran better 'get smart soon' and accept nuclear deal
-
UN experts urge Saudi labour practices switch before World Cup

-
 Oil spikes while stocks slide ahead of US Fed rate decision
Oil spikes while stocks slide ahead of US Fed rate decision
-
US Fed chief's plans in focus as central bank set to hold rates steady

-
 Tuareg rebels vow Mali junta 'will fall', north will be captured
Tuareg rebels vow Mali junta 'will fall', north will be captured
-
German inflation jumps in April as energy costs surge

-
 Was PSG against Bayern the Champions League's greatest ever game?
Was PSG against Bayern the Champions League's greatest ever game?
-
UBS first-quarter profits jump 80% on investment banking

-
 France's 'roadmap' to exit fossil fuels by 2050
France's 'roadmap' to exit fossil fuels by 2050
-
Chelsea captain Millie Bright retires

-
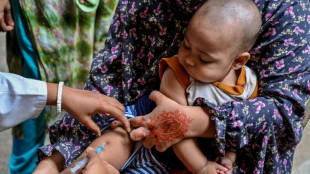 Bangladesh measles outbreak kills over 220 children since March
Bangladesh measles outbreak kills over 220 children since March
-
Finnish lift maker Kone acquires German rival TKE, creating giant

-
 Hungary's Magyar visits Brussels seeking to unblock EU billions
Hungary's Magyar visits Brussels seeking to unblock EU billions
-
Diving robot explores mystery of France's deepest shipwreck

-
 Thai ex-PM Thaksin to be released from prison next month
Thai ex-PM Thaksin to be released from prison next month
-
Welsh rugby great North to hang up his boots

-
 Much-needed rains revive Iraq's fabled Mesopotamian Marshes
Much-needed rains revive Iraq's fabled Mesopotamian Marshes
-
French teen in straw licking case allowed to leave Singapore

-
 EU chief says Kremlin imposing 'digital Iron Curtain' on Russians
EU chief says Kremlin imposing 'digital Iron Curtain' on Russians
-
South Korean court hikes ex-president's sentence for obstructing justice

-
 Adidas reports higher profits but warns of 'volatile' climate
Adidas reports higher profits but warns of 'volatile' climate
-
TotalEnergies first-quarter profits surge amid Middle East war

-
 Sri Lanka government 'temporarily' takes over cricket board
Sri Lanka government 'temporarily' takes over cricket board
-
EU finds Meta failing to keep under-13s off Facebook, Instagram

-
 King Charles to stress UK-US cultural, trade ties in New York
King Charles to stress UK-US cultural, trade ties in New York
-
US judge orders Purdue Pharma to pay billions ahead of bankruptcy

-
 'Jurassic Park' star Sam Neill says cancer-free after gene therapy
'Jurassic Park' star Sam Neill says cancer-free after gene therapy
-
US opioid crisis victims testify at emotional Purdue Pharma hearing

-
 Australian climber on record sea-to-summit Everest bid
Australian climber on record sea-to-summit Everest bid
-
Indian opposition slams Nicobar megaport plan as 'destruction'

-
 Pentagon chief to testify on Iran war, peace efforts stall
Pentagon chief to testify on Iran war, peace efforts stall
-
Anxiety, resentment around AI spur violence against tech's figureheads

-
 Mercedes-Benz profit slides amid cutthroat Chinese market
Mercedes-Benz profit slides amid cutthroat Chinese market
-
Hungary's Magyar to push post-Orban EU reset on Brussels visit

-
 Going online helps Pakistan's women doctors back to work
Going online helps Pakistan's women doctors back to work
-
Wembanyama's Spurs advance in NBA playoffs, 76ers stay alive

-
 Tropical forest loss eases after record year: researchers
Tropical forest loss eases after record year: researchers
-
Tigres edges Nashville in CONCACAF Champions Cup first leg

-
 New Zealand officials reject statue remembering Japan's sex slaves
New Zealand officials reject statue remembering Japan's sex slaves
-
King Charles, Trump toast ties despite Iran tensions

-
 Japan cleaner goes viral with spa-like service for plushies
Japan cleaner goes viral with spa-like service for plushies
-
What we learned from cycling's Spring Classics

-
 Villa, Forest revive European glory days in semi-final showdown
Villa, Forest revive European glory days in semi-final showdown
-
Remarkable, ramshackle Rayo chasing Conference League dream amid chaos

-
 Unbeaten records on the line for Inoue-Nakatani superfight in Tokyo
Unbeaten records on the line for Inoue-Nakatani superfight in Tokyo
-
Cheaper, cleaner electric trucks overhaul China's logistics

-
 Stocks swing, oil edges up with Iran war peace talks stalled
Stocks swing, oil edges up with Iran war peace talks stalled
-
Europe climate report signals rising extremes

-
 Sexual violence in Sudan triggers mental health crisis: UN
Sexual violence in Sudan triggers mental health crisis: UN
-
The loyal, lonely keepers of Sudan's pyramids


Hemogenyx Pharmaceuticals PLC Announces Third Patient Safety
Third Patient Treated with HG-CT-1 CAR-T Therapy Successfully Passes Initial Safety Evaluation
Third Patient Treated with HG-CT-1 CAR-T Therapy Successfully Passes Initial Safety Evaluation
LONDON, UNITED KINGDOM / ACCESS Newswire / September 17, 2025 / Hemogenyx Pharmaceuticals plc is pleased to announce that the third patient has been successfully treated in the ongoing Phase I clinical trial of HG-CT-1, the Company's proprietary Chimeric Antigen Receptor T-cell (CAR-T) therapy for relapsed/refractory acute myeloid leukemia (R/R AML) in adults.
The treatment was well tolerated and met the trial's predefined initial safety criteria. Importantly, early indications of clinical efficacy have been observed. Preliminary assessment shows that original AML cells were not detectable in the patient using standard testing methods. The patient will continue to be monitored in line with the FDA-approved trial protocol to evaluate the achievement of the study's secondary endpoints described below.
Safety data from the first three patients treated at the lowest dose of HG-CT-1 will be submitted to an independent Data Safety Monitoring Board (DSMB) for review. The DSMB will determine whether dose escalation to the next level may proceed.
The Phase I trial is a dose-escalation study designed to evaluate the safety and tolerability of HG-CT-1. In addition to safety, the trial includes several key secondary endpoints:
Assessing the efficacy of HG-CT-1 based on AML-specific response criteria
Evaluating overall survival
Measuring progression-free survival
Determining duration of response in patients demonstrating clinical benefit
Data related to these secondary endpoints, including efficacy, durability, and overall clinical outcomes, will be collected over time through continued follow-up of the treated patient. These secondary endpoints are critical for assessing the potential clinical impact of HG-CT-1 in a patient population with limited remaining treatment options.
Further updates will be provided as the trial progresses.
Dr Vladislav Sandler, CEO & Co-Founder of Hemogenyx Pharmaceuticals, commented:
"The successful treatment of the third patient marks another important milestone for Hemogenyx Pharmaceuticals and for patients battling relapsed or refractory AML. We are encouraged by the favorable safety profile observed to date, together with the early signals of efficacy. These results reinforce the promise of HG-CT-1 as a potential new therapy for one of the most aggressive and intractable forms of leukemia. We remain committed to advancing the clinical development of this therapy to address a critical unmet medical need, while also creating long-term value for our shareholders."
Market Abuse Regulation (MAR) Disclosure
Certain information contained in this announcement would have been inside information for the purposes of Article 7 of Regulation No 596/2014 (as it forms part of UK domestic law by virtue of the European Union (Withdrawal) Act 2018) until the release of this announcement.
Enquiries:
Hemogenyx Pharmaceuticals plc | |
Dr Vladislav Sandler, Chief Executive Officer & Co-Founder | |
Peter Redmond, Director | |
SP Angel Corporate Finance LLP | Tel: +44 (0)20 3470 0470 |
Matthew Johnson, Vadim Alexandre, Adam Cowl | |
Peterhouse Capital Limited | Tel: +44 (0)20 7469 0930 |
Lucy Williams, Duncan Vasey, Charles Goodfellow | |
About Hemogenyx Pharmaceuticals plc
Hemogenyx Pharmaceuticals is a publicly traded company (LSE:HEMO) headquartered in London, with its US operating subsidiaries, Hemogenyx Pharmaceuticals LLC and Immugenyx LLC, located in New York City.
The Company is a clinical stage biopharmaceutical group developing new medicines and treatments to treat blood and autoimmune diseases. Hemogenyx Pharmaceuticals is developing several distinct and complementary product candidates, as well as platform technologies that it uses as engines for novel product development.
This information is provided by RNS, the news service of the London Stock Exchange. RNS is approved by the Financial Conduct Authority to act as a Primary Information Provider in the United Kingdom. Terms and conditions relating to the use and distribution of this information may apply. For further information, please contact [email protected] or visit www.rns.com.
SOURCE: Hemogenyx Pharmaceuticals PLC
View the original press release on ACCESS Newswire
F.Ramirez--AT

