
-
 Saving energy in everyday life or a complete rip-off?
Saving energy in everyday life or a complete rip-off?
-
US sprint star Richardson wins Australia's Stawell Gift in record time

-
 Rockets down Warriors in Curry return, Flagg carries Mavs past Lakers
Rockets down Warriors in Curry return, Flagg carries Mavs past Lakers
-
Artemis mission approaches lunar loop for first flyby since 1972

-
 Israeli rescuers search for missing in building strike, two dead
Israeli rescuers search for missing in building strike, two dead
-
Defiant Iran ramps up attacks after Trump warning

-
 Saudi oasis town adjusts to life in the firing line
Saudi oasis town adjusts to life in the firing line
-
Pogacar stays humble with Monument history beckoning

-
 Real Madrid hoping Champions League magic halts Bayern juggernaut
Real Madrid hoping Champions League magic halts Bayern juggernaut
-
Sputtering Arsenal face test of character in Sporting clash

-
 'Not the Cairo we know': Energy shock from Iran war dims Egypt nights
'Not the Cairo we know': Energy shock from Iran war dims Egypt nights
-
Tokyo, Seoul shares gain, war sends oil higher

-
 Artemis mission headed for first lunar flyby since 1972
Artemis mission headed for first lunar flyby since 1972
-
South Korea president says regrets 'reckless' drones sent to North

-
 Coughlin captures third LPGA title at Aramco Championship
Coughlin captures third LPGA title at Aramco Championship
-
What to know about the Artemis 2 mission's Moon flyby

-
 Mystique of the green jacket endures as Masters looms
Mystique of the green jacket endures as Masters looms
-
In El Salvador's mass trials, 'the innocent pay for the guilty'

-
 Trump makes stark threat to Iran after US airman rescued
Trump makes stark threat to Iran after US airman rescued
-
Texas Gulf Bank, N.A. Appoints Chase Zalman President

-
 Bioz and Vilber Advance Evidence-Driven Scientific Marketing with Custom Publication Integration
Bioz and Vilber Advance Evidence-Driven Scientific Marketing with Custom Publication Integration
-
Artemis astronauts ready for Moon flyby on fifth day of historic mission

-
 Israel renews Lebanon strikes, forces Syria border crossing closed
Israel renews Lebanon strikes, forces Syria border crossing closed
-
Eagle-eyed Spaun snatches Texas Open victory

-
 Brown, Tatum propel Celtics in win over Raptors
Brown, Tatum propel Celtics in win over Raptors
-
Paul battles past Burruchaga to win ATP Houston title

-
 Major sponsors drop Kanye West London gigs as PM voices concern
Major sponsors drop Kanye West London gigs as PM voices concern
-
Inter close in on Serie A title by thumping Roma

-
 Trump makes foul-mouthed threat to Iran after US airman rescued
Trump makes foul-mouthed threat to Iran after US airman rescued
-
Monaco sink Marseille for seventh Ligue 1 win in a row

-
 Inter thump Roma to extend Serie A lead to nine points
Inter thump Roma to extend Serie A lead to nine points
-
Lebanon's Christians mark Easter in solidarity with war-hit south

-
 Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years
Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years
-
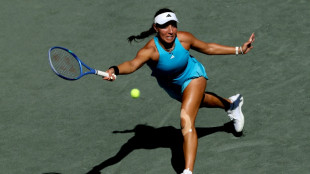
Pegula romps to WTA Charleston Open victory
-
 David six-hitting spree powers Bengaluru to IPL win
David six-hitting spree powers Bengaluru to IPL win
-
Union draw leaves St Pauli stranded in Bundesliga drop zone

-
 UK police arrest protesters near base used by US
UK police arrest protesters near base used by US
-
Trump issues foul-mouthed threat to Iran after US airman rescued

-
 Alcaraz plans to play full clay-court season, get 'socks dirty'
Alcaraz plans to play full clay-court season, get 'socks dirty'
-
'Super Mario Galaxy' blasts off in N. America box office debut

-
 Artemis astronauts begin fifth day on historic Moon mission
Artemis astronauts begin fifth day on historic Moon mission
-
Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup

-
 Trump draws criticism with fiery Easter message on Iran
Trump draws criticism with fiery Easter message on Iran
-
OPEC+ hikes oil production quotas, issues warning

-
 British PM slams London event for booking Kanye West, sponsor quits
British PM slams London event for booking Kanye West, sponsor quits
-
Pogacar wins joint-record third Tour of Flanders

-
 Trump threatens 'hell' for Iran over Strait of Hormuz
Trump threatens 'hell' for Iran over Strait of Hormuz
-
Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash

-
 What we know about the race to rescue downed US airman in Iran
What we know about the race to rescue downed US airman in Iran
-
US commandos went deep into Iran to rescue downed airman: media


Ayrmid Pharma Ltd Announces Additional Positive Results for Omisirge(R) in Treating Severe Aplastic Anemia (SAA) Presented at TANDEM
Omisirge transplantation shows accelerated immune recovery in SAA
New data presented at the 2026 TANDEM Meetings demonstrate that patients with SAA who received Omisirge experienced faster and more robust immune recovery compared with patients receiving alternative transplant graft sources
Omisirge approved by FDA as First Cell Therapy to Treat Severe Aplastic Anemia
Key Highlights of Data Presented at TANDEM:
Rapid neutrophil recovery: Median time to neutrophil engraftment was 10 days following Omisirge transplantation. Faster recovery was associated with higher transplanted CD34⁺ cell doses
Enhanced CD4⁺T-cell reconstitution: At six months post-transplant, CD4⁺ T-cell counts were significantly higher in Omisirge recipients compared with other transplant sources including matched and haploidentical donors. Higher CD4⁺ T-cell recovery correlated with more rapid neutrophil engraftment
Accelerated NK-cell recovery: Natural killer (NK) cell reconstitution occurred early after transplant, with median NK-cell counts at days 30 and 100 exceeding those observed across all comparator graft sources
Omisirge approved by the FDA on December 5, 2025 for SAA
DUBLIN, IE / ACCESS Newswire / February 5, 2026 / Ayrmid Ltd. ("Ayrmid" or the "Company"), the parent company of Gamida Cell Inc., a leader in cell therapy innovation, today announced further positive interim clinical results for Omisirge (Omidubicel-onlv), its advanced stem cell transplant therapy for Severe Aplastic Anemia (SAA). The data showed that patients with SAA who received Omisirge transplants experienced faster and more robust immune recovery compared with patients receiving other transplant graft sources, including HLA-matched donor peripheral blood stem cells, haploidentical donors with post-transplant cyclophosphamide, standard cord blood, and combined cord/haploidentical transplants. The findings come from an ongoing single-center Phase II study conducted at the National Heart, Lung, and Blood Institute (NHLBI) of the National Institutes of Health (NIH) and were presented by investigators from NHLBI/NIH and collaborating institutions at the 2026 Transplantation & Cellular Therapy meetings of ASTCT® and CIBMTR® (TANDEM), taking place from February 4-7, 2026 in Salt Lake City, UT.
Alternative donor hematopoietic cell transplantation (HCT) has historically been limited in SAA by delayed engraftment and high rates of graft rejection. Omisirge is a nicotinamide-expanded cord blood product designed to increase the number of hematopoietic stem and progenitor cells available for transplantation.
Dr. Richard Childs of the National Heart, Lung, and Blood Institute (NHLBI) at the National Institutes of Health (NIH) commented: "In this study, we observed that Omisirge transplantation was associated with faster neutrophil engraftment and earlier recovery of key immune cell populations compared with historical transplant approaches in SAA. These findings suggest that enhanced stem cell dose may translate into improved early immune reconstitution in this high-risk patient population."
Dr. Ronit Simantov, Chief Medical and Scientific Officer of Ayrmid commented: "These results highlight the significant potential of Omisirge as a transplant option for patients with SAA. The enhanced immune recovery observed following Omisirge transplantation may have important clinical implications for patients with SAA, including improved engraftment, reduced risk of graft rejection, and potentially lower susceptibility to infections in the post-transplant period. We remain deeply committed to advancing transformative therapies for patients with serious unmet medical needs."
Omisirge is now approved for treatment in adults and pediatric patients 6 years of age and older with SAA following reduced intensity conditioning. The FDA approval of Omisirge was based on data from the 17-H-0091 study.
About Severe Aplastic Anemia
Severe Aplastic Anemia is a rare, life-threatening hematologic disorder in which the bone marrow fails to produce sufficient blood cells. Stem cell transplantation offers a potential cure; however, many patients lack a matched sibling donor. Ayrmid is advancing therapies to address this unmet medical need.
About Ayrmid Ltd. and Gamida Cell
Ayrmid Ltd. is the parent company of Gamida Cell Inc., a pioneering cell therapy company developing novel treatments designed to turn cells into powerful therapeutics. Gamida Cell Inc. currently has two FDA approved products on the market in the US, namely Omisirge® (please see the current full Prescribing Information, including boxed warning, here) and APHEXDA® (please see the current full Prescribing Information here). Gamida Cell operates as a wholly owned subsidiary of Ayrmid Limited, a UK entity. For additional information, please visit www.gamida-cell.com or follow Gamida Cell on LinkedIn, X, Facebook or Instagram.
Contacts: Media, Investors / Business Development: [email protected]
This information is provided by RNS, the news service of the London Stock Exchange. RNS is approved by the Financial Conduct Authority to act as a Primary Information Provider in the United Kingdom. Terms and conditions relating to the use and distribution of this information may apply. For further information, please contact [email protected] or visit www.rns.com.
SOURCE: Ayrmid Pharma Ltd
View the original press release on ACCESS Newswire
M.Robinson--AT
