
-
 Fitzpatrick tries to balance goals ahead of Masters
Fitzpatrick tries to balance goals ahead of Masters
-
Trump branded 'crazy' over apocalyptic Iran threats

-
 Vance hails Orban as 'model' for Europe in pre-election Hungary visit
Vance hails Orban as 'model' for Europe in pre-election Hungary visit
-
McIlroy starting with Young, Howell in Masters repeat bid

-
 Picasso's 'Guernica' at heart of battle in Spain over location
Picasso's 'Guernica' at heart of battle in Spain over location
-
Isak named in Liverpool squad for PSG clash after long injury absence

-
 Young says rise up rankings gives him belief for Masters
Young says rise up rankings gives him belief for Masters
-
Artemis II crew snaps historic Earthset photo on way home

-
 Seixas climbs to victory to extend Basque Tour lead
Seixas climbs to victory to extend Basque Tour lead
-
Oil rises, stocks fall ahead of Trump's Iran deadline

-
 With Legos, trolling and Twain, Iran pushes war narrative on social media
With Legos, trolling and Twain, Iran pushes war narrative on social media
-
Rahm confident of playing '27 Ryder Cup and DP World Tour

-
 French couple leave Iran after more than three years in detention
French couple leave Iran after more than three years in detention
-
NASA releases picture of 'Earthset' shot by Artemis crew

-
 Major dreams and Middle East War in Fleetwood's Masters thoughts
Major dreams and Middle East War in Fleetwood's Masters thoughts
-
Trump warns 'whole civilization will die' in Iran if ultimatum expires

-
 Sinner and Alcaraz start fast on Monte Carlo clay in race for No.1
Sinner and Alcaraz start fast on Monte Carlo clay in race for No.1
-
UK government blocks Kanye West from London music fest

-
 Oil rises, stocks fall as Trump's Iran deadline looms
Oil rises, stocks fall as Trump's Iran deadline looms
-
Graft trial of Spanish PM's ex-top aide begins

-
 French high-speed train slams into truck, killing TGV driver
French high-speed train slams into truck, killing TGV driver
-
Kanye West offers to meet UK Jewish community amid music fest row

-
 Key infrastructure in Iran hit ahead of Trump deadline
Key infrastructure in Iran hit ahead of Trump deadline
-
Sinner keeps run going by crushing Humbert in Monte Carlo

-
 Ex-footballer Barton denies assault near golf club
Ex-footballer Barton denies assault near golf club
-
Barca's Flick to defend 'emotional' teen Yamal against criticism

-
 Two children among 12 dead in fresh Ukraine, Russia strikes
Two children among 12 dead in fresh Ukraine, Russia strikes
-
PSG wary of wounded Liverpool ahead of European showdown

-
 Ex-Arsenal midfielder Ramsey retires at 35
Ex-Arsenal midfielder Ramsey retires at 35
-
Conte says Italian federation should consider him for coach's job

-
 Makhmudov hails heavyweight 'legend' Fury ahead of London clash
Makhmudov hails heavyweight 'legend' Fury ahead of London clash
-
Juve's Vlahovic suffers latest injury setback

-
 Australian cricket great David Warner charged with drink-driving: reports
Australian cricket great David Warner charged with drink-driving: reports
-
McKeown edges O'Callaghan, dominant Pallister wins 400m freestyle at Australian Open

-
 Oil, stocks rise as Trump's Iran deadline looms
Oil, stocks rise as Trump's Iran deadline looms
-
Gunman killed, 2 wounded in shootout outside Israel's Istanbul consulate

-
 US fund Pershing Square launches takeover bid for Universal Music
US fund Pershing Square launches takeover bid for Universal Music
-
Train driver killed, two critically injured as French TGV collides with truck

-
 Maguire signs one-year Man Utd contract extension
Maguire signs one-year Man Utd contract extension
-
New strikes in Tehran as deadline looms for Trump threat to infrastructure

-
 France's Sarkozy says 'innocent' at trial over Libya funding
France's Sarkozy says 'innocent' at trial over Libya funding
-
In Algeria, Saint Augustine's city anticipates Pope Leo's visit

-
 Veteran Lawes eyes England return after signing for Sale
Veteran Lawes eyes England return after signing for Sale
-
Nepal vows action against trekker rescue scam

-
 Oil prices rally, stocks edge up after Trump's latest Iran threat
Oil prices rally, stocks edge up after Trump's latest Iran threat
-
'Charlie's Angels' stars reunite for show's 50th anniversary

-
 Laughter, tears: historic day for astronaut Jenni Gibbons in Houston
Laughter, tears: historic day for astronaut Jenni Gibbons in Houston
-
Former Wallaby 'Iceman' Foley to retire

-
 Croatia finally landmine-free 30 years after war, but wounds remain
Croatia finally landmine-free 30 years after war, but wounds remain
-
Taiwan opposition leader in China: what you need to know


Regentis Biomaterials Commences Trading on NYSE: Set to Transform Cartilage Repair Market with Off-the-Shelf Regenerative Product
Lead product GelrinC®, a hydrogel synchronized erosion and resorbable implant, the only restorative product for knee cartilage repair, is a breakthrough effective and economical procedure to address a large unmet need
GelrinC is approved for knee cartilage repair in the European Union and is currently at midpoint in a pivotal FDA trial for the same indication to address a U.S. market of more than 470,000 potential cases annually
Upcoming expected catalysts include commercial launch in Europe and submission to FDA for approval in U.S.
HERZLIYA, IL / ACCESS Newswire / December 8, 2025 / Regentis Biomaterials Ltd., ("Regentis" or the "Company") (NYSE American:RGNT), a late clinical stage regenerative medicine company focused on innovative tissue repair solutions, is now trading on the NYSE American under the symbol "RGNT".
Regentis' lead product, GelrinC®, is a cell-free, off-the-shelf hydrogel synchronized erosion and resorbable implant for the treatment of painful injuries to focal articular knee cartilage. As an innovative regenerative medical product, GelrinC offers an unprecedented solution that gives surgeons and payers an off the shelf, ready to use, simple-to-perform, reliable, and cost-effective procedure that provides patients with a single, 10-minute procedure, faster recovery, sustained pain relief, and functional improvement for more than 4 years, based on clinical study results to date. No effective off-the-shelf, ready to use treatment for focal knee cartilage defects is currently available on the market.
GelrinC received CE Mark approval in the European Union where efforts toward commercial launch will start in late 2025 and into 2026, upon engaging a distribution partner. Regentis is currently conducting a pivotal U.S. Food and Drug Administration (FDA) study, which has completed over 50% enrollment.
Upon launch in Europe and approval in the U.S., Regentis expects rapid market adoption driven by the benefits delivered across the value chain to patients, surgeons, and payers.
"With our listing on the NYSE, we are very well positioned to complete the pivotal study and FDA regulatory work for GelrinC in the U.S. and offer a simple and effective regenerative medicine solution for people suffering from painful knee cartilage injuries. Using GelrinC, patients restore their own cartilage for a fresh quality new start," stated Regentis' Executive Chairman, Dr. Ehud Geller. "In the coming quarters, we expect several value drivers including commercial launch in Europe and progress on our FDA trial toward submission of our Premarket Approval (PMA)."
Data from a Phase 2 study in Europe demonstrated the quick and simple GelrinC implantation procedure took only 10 minutes, compared to up to many weeks for competing procedures. With GelrinC, recovery takes approximately 2 weeks, compared to 6 weeks for cellular treatments. The current gold standard treatment, microfracture, which involves creating small punctures in the bone, offers relief on average for 9 to 12 months while GelrinC has shown not only a therapeutic duration of 4 years so far, but also continues to show improvement in pain scores throughout this period.
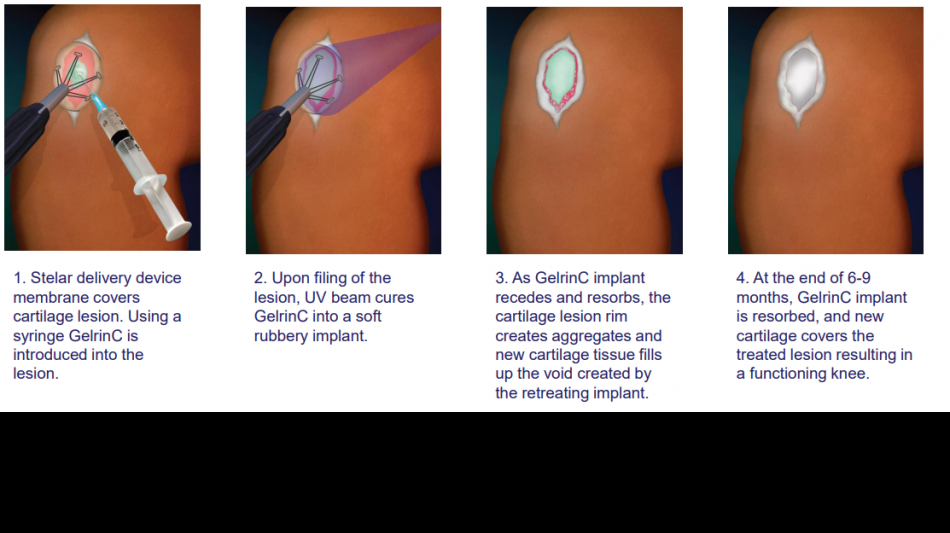
Gelrin is a platform technology that Regentis plans to also develop for other cartilage injuries related to the ankle, wrist, and elbow, as well as the treatment of moderate osteoarthritis. Regentis has 35 granted patents and 3 more pending covering compositions, delivery device, surgical, and manufacturing features.
Ronen Kantor of Amit, Pollak, Matalon & Co. has served as Regentis' legal counsel throughout its IPO process.
About Regentis Biomaterials
Regentis Biomaterials Ltd is a regenerative medicine company dedicated to developing innovative tissue repair solutions that restore health and enhance quality of life. With an initial focus on orthopedic treatments, Regentis' Gelrin platform technology, based on synchronized, degradable hydrogel implants, regenerates damaged or diseased tissue including inflamed cartilage and bone. Regentis' lead product GelrinC, is a cell-free, off-the-shelf hydrogel that is eroded and resorbed in the knee, allowing the surrounding cells to regenerate the cartilage in a controlled and synchronous process. GelrinC addresses a market of 470,000 cases for cartilage knee repair annually in the U.S. where no off-the-shelf treatment is available.
Forward Looking Statements
This press release contains "forward-looking statements" that are subject to substantial risks and uncertainties. All statements, other than statements of historical fact, contained in this press release are forward-looking statements. Forward-looking statements contained in this press release may be identified by the use of words such as "anticipate," "believe," "contemplate," "could," "estimate," "expect," "intend," "seek," "may," "might," "plan," "potential," "predict," "project," "target," "aim," "should," "will" "would," or the negative of these words or other similar expressions, although not all forward-looking statements contain these words, and include the expected start of trading of the Ordinary Shares on the NYSE American LLC, the expected use of proceeds, and the expected date of closing of the Offering. Forward-looking statements are based on Regentis' current expectations and are subject to inherent uncertainties, risks and assumptions that are difficult to predict. Further, certain forward-looking statements are based on assumptions as to future events that may not prove to be accurate. For a more detailed description of the risks and uncertainties affecting Regentis, reference is made to the Company's reports filed from time to time with the Securities and Exchange Commission ("SEC"), including, but not limited to, the risks detailed in the section titled "Risk Factors" in the final prospectus related to the public offering filed with the SEC. Forward-looking statements contained in this announcement are made as of this date, and Regentis undertakes no duty to update such information except as required under applicable law.
Contact:
SOURCE: Regentis Biomaterials Ltd
View the original press release on ACCESS Newswire
W.Moreno--AT

