-
 Napoli threaten absent Lukaku with disciplinary action
Napoli threaten absent Lukaku with disciplinary action
-
German whale saga continues as struggling animal beached again

-
 Chelsea's Cucurella laments 'instability' caused by Maresca exit
Chelsea's Cucurella laments 'instability' caused by Maresca exit
-
'Iran will be at World Cup' and play in US, FIFA's Infantino tells AFP

-
 Stocks rise on peace hopes, oil flat
Stocks rise on peace hopes, oil flat
-
Senegal enacts law doubling penalty for same-sex relations

-
 De Zerbi 'agrees in principle' to become new Tottenham boss - reports
De Zerbi 'agrees in principle' to become new Tottenham boss - reports
-
Trump says other countries should 'just take' the Strait of Hormuz

-
 Russian oil tanker docks in Cuba after US blockade relief
Russian oil tanker docks in Cuba after US blockade relief
-
Next days in Iran war will be 'decisive': Pentagon chief

-
 Indonesia rations fuel as prices soar over Mideast war
Indonesia rations fuel as prices soar over Mideast war
-
How Middle East war is driving up shipping costs

-
 Russian tanker brings oil to Cuba as US eases blockade
Russian tanker brings oil to Cuba as US eases blockade
-
Asia to be hit hardest by Iran war energy crisis: Kpler to AFP

-
 Huawei reports slowing revenue growth in 2025
Huawei reports slowing revenue growth in 2025
-
Sexualised deepfakes targeting actress spur German '#MeToo' moment

-
 Australia head to World Cup on a high after crushing Curacao 5-1
Australia head to World Cup on a high after crushing Curacao 5-1
-
Italy fertility rate fell to new low of 1.14 in 2025

-
 Pakistan cricketer Zaman gets two-match PSL ban for ball tampering
Pakistan cricketer Zaman gets two-match PSL ban for ball tampering
-
Oil prices rise, stocks mixed on Iran war uncertainty

-
 In Beirut's largest stadium, displaced people with disabilities face 'ordeal'
In Beirut's largest stadium, displaced people with disabilities face 'ordeal'
-
Deposed and detained: Niger president's fate unclear nearly three years on

-
 Newcastle say no manager change 'at the moment'
Newcastle say no manager change 'at the moment'
-
Newly-hatched rare Indian bustard chick gets 50-strong guard

-
 Stranded whale frees itself again off German coast
Stranded whale frees itself again off German coast
-
Archaeologists forced by Mideast war to cut short Iraq digs

-
 Stranded whale frees itself again off German coast and disappears
Stranded whale frees itself again off German coast and disappears
-
Thailand's king endorses new cabinet

-
 China bans entombing cremated remains in empty flats
China bans entombing cremated remains in empty flats
-
Calls grow for 15-year-old Suryavanshi to make India bow

-
 Stocks slip, oil swings after report says Trump willing to end war
Stocks slip, oil swings after report says Trump willing to end war
-
Pakistan cricketer Naseem fined record $71,500 for minister criticism

-
 China teen diving prodigy nearly retired after 'reaching mental limit'
China teen diving prodigy nearly retired after 'reaching mental limit'
-
Myanmar junta chief elected vice-president

-
 Russian tanker set to deliver oil to crisis-hit Cuba
Russian tanker set to deliver oil to crisis-hit Cuba
-
Iran fires missiles across Middle East as Trump threatens oil hub

-
 Indonesia summons Google, Meta for 'not complying' with teen social media ban: minister
Indonesia summons Google, Meta for 'not complying' with teen social media ban: minister
-
Wembanyama at the double as Spurs beat Bulls

-
 Australia investigates tech giants over social media ban breaches
Australia investigates tech giants over social media ban breaches
-
Hindu devotional clubbing sways India's youth

-
 Oil slips, stocks rise as report says Trump willing to end war
Oil slips, stocks rise as report says Trump willing to end war
-
Mind games: How football stars are fuelling chess boom

-
 Indonesia trims meals programme: what next?
Indonesia trims meals programme: what next?
-
'A very big deal': Canadian astronaut reflects on historic Moon mission

-
 US pro table tennis league blasts niche sport into spotlight
US pro table tennis league blasts niche sport into spotlight
-
Iran defiant as Trump threatens to destroy oil island

-
 UPDATE: Lt. General (Ret.) Keith Kellogg Joins Powerus Board of Advisors to Shape the Future of U.S. Autonomous Drone Defense
UPDATE: Lt. General (Ret.) Keith Kellogg Joins Powerus Board of Advisors to Shape the Future of U.S. Autonomous Drone Defense
-
Inc. Names iTrustCapital to Its 2026 List of the Fastest-Growing Private Companies in the Pacific

-
 HighLevel Ranked No. 13 on Inc.'s 2026 Regionals: Southwest List
HighLevel Ranked No. 13 on Inc.'s 2026 Regionals: Southwest List
-
Centennial and Lincoln Launch "Centennial, powered by Lincoln" National Retail Operating Platform


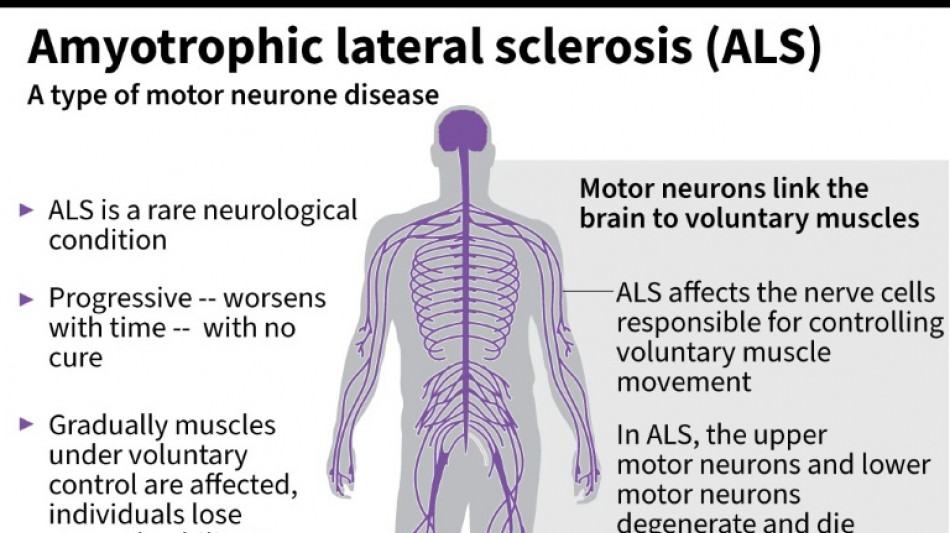
Every month counts: European ALS patients want new drugs
Olivier Goy is running out of time.
The French entrepreneur was diagnosed in 2020 with amyotrophic lateral sclerosis (ALS) -- the incurable neurodegenerative disease that normally claims the lives of patients within three to five years.
There are new treatments that have given patients hope of being able to extend their lives by an invaluable few months, but the approval process in Europe is taking time, infuriating desperate patients.
"When you are certain to die soon, patients and some doctors are ready to take some risks," Goy told AFP.
In response to the lack of new treatments in his native France, the founder of the fintech start-up October spends 3,000 euros ($3,180) every month to buy the ingredients to make his own drugs.
ALS, also known as Lou Gehrig's disease, attacks the motor nerve cells in the brain and spinal cord, progressively paralysing muscles until patients cannot walk, eat, speak or breathe.
Around one in 10,000 people have the disease in the EU, according to the European Medicines Agency.
The drug Riluzole, which has been available in Europe and the UK since the 1990s, is capable of prolonging the lives of patients by around three months.
But otherwise, no new treatment has been approved in Europe for more than two decades.
- 'First hope in 20 years' -
A new treatment called AMX0035 was given the green light in the United States and Canada last year.
"It is the first hope we have had in 20 years: the first drug which is aimed at everyone and which had results" suggesting up to six months in added life expectancy, said Sabine Turgeman, head of the French Association for Research into ALS.
But the extent of the benefits of AMX0035 remains unclear. The US Food and Drug Administration approved the drug, sold under the name Relyvrio, based on the results of a single Phase 2 trial that involved just 137 participants.
The drug's developer, Amylyx Pharmaceuticals, is conducting larger, more comprehensive trials, with results expected in 2024.
Amylyx said earlier this month that the European Union's drug watchdog EMA is reviewing its submission for approval and it expects a decision in the first half of this year.
But for those with the disease, every delay represents a significant amount of the time they have left.
"It's not going fast enough," Turgeman said. "This disease is not on bureaucratic time".
For European patients who cannot afford to import their own ingredients like Goy, the only way to get access to new treatments is to join a clinical trial.
But such trials have very specific criteria for selection -- and even if a patient gets in, there is a chance they will be in the group given a placebo.
- 'Totally abandoned' -
Given how swiftly the disease progresses, patients and families are pressing for more options.
"We feel totally abandoned," said Sophie Garofalo, whose brother was diagnosed with ALS five years ago.
His family tried to enter him into clinical trials, "but either he does not meet the criteria, or the trials have already started," she said.
"He is ready to take anything, try everything".
French pharmaceutical company AB Science is developing another potential treatment using the drug masitinib, which initial results suggest could add months to the lives of patients.
The firm's CEO Alain Moussy said that because "time is very limited" for ALS patients, there should be more flexibility in the approval system.
"What degree of risk should be taken? That's for the health agencies to answer -- but they can guided by policymakers and patients," he said.
E.Rodriguez--AT




