-
 Arsenal's Raya hailed as 'world's best keeper' after denying Sporting
Arsenal's Raya hailed as 'world's best keeper' after denying Sporting
-
Bayern's Kompany praises 'special' Neuer display in win at Real Madrid

-
 Diaz, Kane give Bayern vital Champions League win at Real
Diaz, Kane give Bayern vital Champions League win at Real
-
Havertz strikes late as Arsenal steal Champions League advantage against Sporting

-
 Pakistan makes last-minute bid to avert Trump threat to destroy Iran
Pakistan makes last-minute bid to avert Trump threat to destroy Iran
-
Artemis II crew basks in glow of lunar flyby en route to Earth

-
 Global stocks mostly fall ahead of Trump's deadline for Iran
Global stocks mostly fall ahead of Trump's deadline for Iran
-
Trump weighs plea for Iran deadline extension

-
 Artemis and ISS astronauts share celestial call
Artemis and ISS astronauts share celestial call
-
Former Romania coach Lucescu dies aged 80

-
 'Nice to get a 2nd chance': Slot tips Liverpool to bounce back against PSG
'Nice to get a 2nd chance': Slot tips Liverpool to bounce back against PSG
-
Iran says ready for anything after Trump warns 'whole civilization will die'

-
 French couple head home after more than three years in Iranian jail
French couple head home after more than three years in Iranian jail
-
Jaiswal, Sooryavanshi fire Rajasthan to win in rain-hit IPL clash

-
 Extra Masters security eases anxiety battle for Woodland
Extra Masters security eases anxiety battle for Woodland
-
Atletico's Simeone hails 'exemplary' departing Griezmann

-
 Relaxed McIlroy finds new challenges after Masters win
Relaxed McIlroy finds new challenges after Masters win
-
Russia, China veto UN resolution on reopening Strait of Hormuz

-
 Indigenous groups demand greater land protection in Brazil protest
Indigenous groups demand greater land protection in Brazil protest
-
Fitzpatrick tries to balance goals ahead of Masters

-
 Trump branded 'crazy' over apocalyptic Iran threats
Trump branded 'crazy' over apocalyptic Iran threats
-
Vance hails Orban as 'model' for Europe in pre-election Hungary visit

-
 McIlroy starting with Young, Howell in Masters repeat bid
McIlroy starting with Young, Howell in Masters repeat bid
-
Picasso's 'Guernica' at heart of battle in Spain over location

-
 Isak named in Liverpool squad for PSG clash after long injury absence
Isak named in Liverpool squad for PSG clash after long injury absence
-
Young says rise up rankings gives him belief for Masters

-
 Artemis II crew snaps historic Earthset photo on way home
Artemis II crew snaps historic Earthset photo on way home
-
Seixas climbs to victory to extend Basque Tour lead

-
 Oil rises, stocks fall ahead of Trump's Iran deadline
Oil rises, stocks fall ahead of Trump's Iran deadline
-
With Legos, trolling and Twain, Iran pushes war narrative on social media

-
 Rahm confident of playing '27 Ryder Cup and DP World Tour
Rahm confident of playing '27 Ryder Cup and DP World Tour
-
French couple leave Iran after more than three years in detention

-
 NASA releases picture of 'Earthset' shot by Artemis crew
NASA releases picture of 'Earthset' shot by Artemis crew
-
Major dreams and Middle East War in Fleetwood's Masters thoughts

-
 Trump warns 'whole civilization will die' in Iran if ultimatum expires
Trump warns 'whole civilization will die' in Iran if ultimatum expires
-
Sinner and Alcaraz start fast on Monte Carlo clay in race for No.1

-
 UK government blocks Kanye West from London music fest
UK government blocks Kanye West from London music fest
-
Oil rises, stocks fall as Trump's Iran deadline looms

-
 Graft trial of Spanish PM's ex-top aide begins
Graft trial of Spanish PM's ex-top aide begins
-
French high-speed train slams into truck, killing TGV driver

-
 Kanye West offers to meet UK Jewish community amid music fest row
Kanye West offers to meet UK Jewish community amid music fest row
-
Key infrastructure in Iran hit ahead of Trump deadline

-
 Sinner keeps run going by crushing Humbert in Monte Carlo
Sinner keeps run going by crushing Humbert in Monte Carlo
-
Ex-footballer Barton denies assault near golf club

-
 Barca's Flick to defend 'emotional' teen Yamal against criticism
Barca's Flick to defend 'emotional' teen Yamal against criticism
-
Two children among 12 dead in fresh Ukraine, Russia strikes

-
 PSG wary of wounded Liverpool ahead of European showdown
PSG wary of wounded Liverpool ahead of European showdown
-
Ex-Arsenal midfielder Ramsey retires at 35

-
 Conte says Italian federation should consider him for coach's job
Conte says Italian federation should consider him for coach's job
-
Makhmudov hails heavyweight 'legend' Fury ahead of London clash


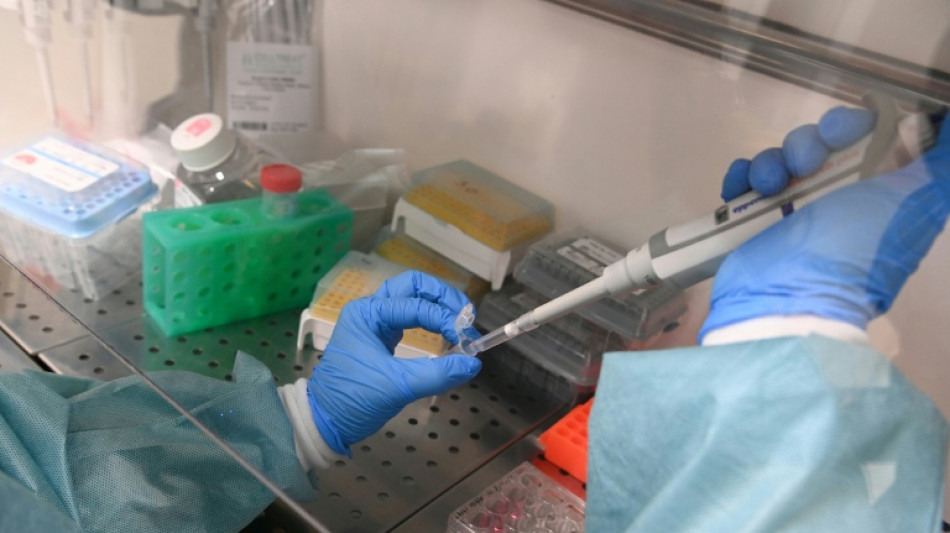
Moderna says US refusing to review mRNA-based flu shot
Vaccine manufacturer Moderna said Tuesday the US Food and Drug Administration was refusing to review an application for its first mRNA-based flu shot.
The move comes as the current iteration of the FDA has called for a reconsideration of approval procedures for certain vaccines, including for influenza -- proposed federal policy changes under President Donald Trump that have triggered widespread alarm among public health and medical professionals.
Moderna, a US company, said the agency's top vaccine regulator, Vinay Prasad, wrote in a letter that Moderna's clinical trial was not "adequate and well-controlled," and had not tested its experimental shot against the best product on the market.
In the large trial Moderna had compared its new vaccine with Fluarix, an approved flu shot from the company GSK.
Moderna said the rejection was "inconsistent with previous written communications" with the FDA branch that regulates biological products, including vaccines, known by the acronym CBER.
The decision "did not identify any safety or efficacy concerns with our product" and "does not further our shared goal of enhancing America's leadership in developing innovative medicines," Moderna CEO Stephane Bancel said in the statement.
"It should not be controversial to conduct a comprehensive review of a flu vaccine submission that uses an FDA-approved vaccine as a comparator in a study that was discussed and agreed on with CBER prior to starting."
Moderna said it had received a Refusal-to-File letter, which would indicate the application did not meet the requirements for substantive review. The company said it had requested a meeting to discuss the decision.
The letter did not identify any safety or efficacy concerns regarding the mRNA vaccine, said Moderna, which added the shot was accepted for review in the European Union, Canada and Australia.
During his first term Trump called mRNA technology a "modern-day miracle."
It was used during the Covid-19 pandemic to swiftly develop an immunization against the fast-spreading illness, and was credited with saving millions of lives.
Vaccines against Covid-19 are considered safe and effective by global health authorities, and protect against the most severe forms of the infection.
But in his second term, Trump has appointed Robert F. Kennedy Jr as his health chief, and the long-time vocal vaccine skeptic has spent the past year reshaping federal health agencies in his image.
That has meant spreading false information and sowing doubt about the safety and efficacy of vaccines widely known to be safe, and upending the pediatric immunization schedule.
Notably, Kennedy cut off federal research grants that funded mRNA development.
F.Wilson--AT
