
-
 Japan rides box office boom into Cannes
Japan rides box office boom into Cannes
-
Trump arrives in China for superpower summit with Xi

-
 UK's Catherine on first official foreign trip since cancer diagnosis
UK's Catherine on first official foreign trip since cancer diagnosis
-
British scientists among winners of top Spanish award

-
 Mbappe can show 'commitment' to Real Madrid: Arbeloa
Mbappe can show 'commitment' to Real Madrid: Arbeloa
-
Chinese tech giant Alibaba posts profit drop amid AI drive

-
 King Charles lays out Starmer's agenda as PM fights for survival
King Charles lays out Starmer's agenda as PM fights for survival
-
Japan suspend Eddie Jones for verbally abusing officials

-
 England drop Crawley for 1st Test against New Zealand
England drop Crawley for 1st Test against New Zealand
-
Stocks rise ahead of US-China summit as Iran talks stall

-
 One trip, one ticket: New EU rules aim to ease train travel
One trip, one ticket: New EU rules aim to ease train travel
-
SoftBank profit quadruples to $32 bn on AI investments

-
 Africa must drop 'victim mentality': mogul Tony Elumelu
Africa must drop 'victim mentality': mogul Tony Elumelu
-
'Ungovernable' Britain? Once-stable politics in freefall

-
 China tech giant Tencent sees Q1 profit jump after AI bets
China tech giant Tencent sees Q1 profit jump after AI bets
-
Nissan expects return to profit after huge loss

-
 World Cup broadcast deadlock ends up in Indian court
World Cup broadcast deadlock ends up in Indian court
-
Asian stocks mixed on US-Iran impasse, AI setbacks

-
 Besieged Starmer seeks to heal Labour divisions in King's Speech
Besieged Starmer seeks to heal Labour divisions in King's Speech
-
After winter storms, fires now threaten Portugal's forests

-
 Philippine senator seeks military support to block ICC drug war arrest
Philippine senator seeks military support to block ICC drug war arrest
-
UK's Catherine on first official foreign trip since cancer revelation

-
 'Short of blue-collar workers': Ukraine's battle for labour
'Short of blue-collar workers': Ukraine's battle for labour
-
'Don't understand it, but it looks fun': cricket bowls Japan over

-
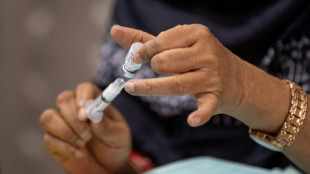 Poor planning fuels Bangladesh contraceptive crisis
Poor planning fuels Bangladesh contraceptive crisis
-
Fugitive financier sought in Malaysian fund scandal seeks Trump's pardon

-
 World Cup comes to 'Soccer Town USA,' but locals priced out
World Cup comes to 'Soccer Town USA,' but locals priced out
-
Don't mention the war: Tucson prepares to welcome Team Iran for World Cup

-
 Hosting World Cup evokes powerful memories for Mexico, and raises expectations
Hosting World Cup evokes powerful memories for Mexico, and raises expectations
-
AI rivalry overshadows push for guardrails at Xi-Trump talks: experts

-
 Asian stocks fall on US-Iran impasse, AI setbacks
Asian stocks fall on US-Iran impasse, AI setbacks
-
Wembanyama leads Spurs to brink as Timberwolves routed

-
 Ronaldo left waiting for Saudi title after goalkeeping gaffe
Ronaldo left waiting for Saudi title after goalkeeping gaffe
-
'Not my son's fault': The women bearing the children of Sudan's war rapes

-
 'I applied to be pope': Losing grip on reality while using ChatGPT
'I applied to be pope': Losing grip on reality while using ChatGPT
-
EU to ease train travel with one journey, one ticket rules

-
 Quick bowler Brown left out of Australia T20 World Cup squad
Quick bowler Brown left out of Australia T20 World Cup squad
-
Los Angeles stadium undergoes World Cup facelift

-
 Pacific nation Nauru to change name in break from colonial past
Pacific nation Nauru to change name in break from colonial past
-
Messi still highest-paid player in MLS

-
 Paramount defends Warner bid amid California probe
Paramount defends Warner bid amid California probe
-
5E Advanced Materials Reports Key Fiscal Q3 2026 Commercial and Operational Milestones

-
 QumulusAI Establishing Corporate Headquarters in Georgia Tech's Tech Square
QumulusAI Establishing Corporate Headquarters in Georgia Tech's Tech Square
-
Veterans With Tax Debt May Qualify for Special IRS Relief - Clear Start Tax Highlights Programs Many Service Members Overlook

-
 Aptevo Therapeutics Provides a 1Q26 Business Update; RAINIER on Track for 2026 Completion and Phase 2 Dose Selection
Aptevo Therapeutics Provides a 1Q26 Business Update; RAINIER on Track for 2026 Completion and Phase 2 Dose Selection
-
Bloomia Holdings, Inc. Announces March 31, 2026 Financial Results

-
 Xenetic Biosciences Reports First Quarter 2026 Financial Results and Highlights Continued Advancement of DNase Oncology Platform
Xenetic Biosciences Reports First Quarter 2026 Financial Results and Highlights Continued Advancement of DNase Oncology Platform
-
MIRA Pharmaceuticals Announces Acceptance of Peer-Reviewed SKNY-1 Manuscript Highlighting Oral Obesity and Nicotine Addiction Drug Candidate

-
 SMX And the Plastic Reset: How Verified Recycling May Determine the Future Cost of Modern Life
SMX And the Plastic Reset: How Verified Recycling May Determine the Future Cost of Modern Life
-
The White House Names Peter Arnell as U.S. Chief Brand Architect within the National Design Studio


Aptevo Therapeutics Provides a 1Q26 Business Update; RAINIER on Track for 2026 Completion and Phase 2 Dose Selection
Mipletamig continues to perform, delivers 87% clinical benefit and 81% remission in frontline AML, with no CRS in frontline patients
Executive leadership transition complete. Jeff Lamothe appointed CEO; Marvin White assumes Executive Chair
$60 million equity line of credit enhances financial flexibility and extends access to capital through achievement of key catalysts into 2029
SEATTLE, WA / ACCESS Newswire / May 13, 2026 / Aptevo Therapeutics Inc. (Nasdaq:APVO), a clinical-stage biotechnology company focused on developing novel immune-oncology therapeutics based on its proprietary ADAPTIR™ and ADAPTIR-FLEX™ platform technologies, today provided a business update.
"Aptevo is entering a defining period as a company, supported by growing clinical momentum, a committed leadership team, and a pipeline with the potential to create significant long-term value," said Jeff Lamothe, President and Chief Executive Officer of Aptevo. "Mipletamig continues to generate compelling frontline AML data, combining strong remission activity with a differentiated safety profile with the potential as an important addition to standard-of-care therapy. At the same time, we are continuing to advance our next generation of multispecific immunotherapy programs, including our emerging trispecific candidates, which are designed to address complex solid tumors through differentiated mechanisms and targeted immune modulation. We believe our ADAPTIR and ADAPTIR-FLEX platforms position Aptevo to expand into multiple high-value areas of oncology innovation. With meaningful clinical and strategic milestones ahead, financial flexibility to support execution, and increasing validation of multispecific approaches throughout the industry, we believe we are building from an increasingly strong foundation for future growth and value creation."
RAINIER trial on track for completion and Phase 2 dose selection by year end.
Mipletamig continues to generate strong data in frontline acute myeloid leukemia (AML) in combination with venetoclax + azacitidine. Across 31 evaluable patients (includes data through RAINIER Cohort 5, plus 4 patients from the previously completed dose expansion trial), the data has demonstrated continued efficacy, including:
87% clinical benefit rate* that demonstrates broad anti-leukemia activity and blast reduction across response categories
81% CR or CRi (remission), compared to 66.4% in the Phase 3 VIALE-A trial**
65% achieved CR (complete remission), compared to 37% in the Phase 3 VIALE-A trial**
No cytokine release syndrome (CRS), a common and often dose-limiting toxicity associated with similar therapies, has been observed in frontline patients to date
The data also show that 52% of patients who achieved CR/CRi had blast reductions that reached the important measurable residual disease-negative level, a result that is typically associated with stronger, more durable responses.
As the dataset continues to expand, efficacy and safety outcomes continue to deliver favorable results, further supporting mipletamig's potential in the frontline setting.
*Clinical benefit rate: complete remission (CR), complete remission with incomplete hematologic recovery (CRi), and partial remission (PR)
**Phase 3 VIALE-A trial evaluating venetoclax plus azacitidine in frontline intent-to-treat AML patients who were ineligible for intensive induction chemotherapy, the reported composite CR/CRi rate was 66.4%, and the CR rate was 37% (DiNardo et al., New England Journal of Medicine, 2020).
Completed Leadership Transitions; Company Poised for a Defining Year
Aptevo entered 2026 with purposeful momentum, highlighted by a planned executive leadership transition designed to support the company's next phase of growth. Jeff Lamothe was appointed President and Chief Executive Officer, while Marvin White transitioned to Executive Chair. The move reflects continuity in strategy while positioning the organization for focused execution across clinical development, capital strategy, and long-term value creation.
Q1 2026 Cash Position
Aptevo had cash and cash equivalents totaling $14.5 million as of March 31, 2026. During the first quarter of 2026, the company raised $0.9 million, net, under the company's Standby Equity Purchase Agreements (SEPAs) with Yorkville. For additional APVO financial information and complete access to the company's filings, click here.
Enhanced Financial Flexibility Supports Upcoming Catalysts
During the quarter, Aptevo secured a $60 million SEPA, providing meaningful access to capital and extending financial flexibility as the company advances toward planned milestones. Management believes the facility better positions Aptevo to execute strategically, support ongoing development programs, and approach future opportunities from a position of greater strength.
About Aptevo Therapeutics
Aptevo Therapeutics Inc. (Nasdaq: APVO) is a clinical-stage biotechnology company focused on developing novel bispecific and trispecific immunotherapies for the treatment of cancer. The company has two clinical candidates. Mipletamig is currently being evaluated in RAINIER, a two-part Phase 1b/2 trial for the treatment of frontline acute myeloid leukemia in combination with standard-of-care venetoclax + azacitidine. Mipletamig has received orphan drug designation ("orphan status") for AML according to the Orphan Drug Act. ALG.APV-527, a bispecific conditional 4-1BB agonist active only upon simultaneous binding to 4-1BB and 5T4, is being co-developed with Alligator Bioscience and was most recently evaluated in a Phase 1 clinical trial for the treatment of multiple solid tumor types likely to express 5T4. The company has six preclinical candidates designed to target a range of solid tumors. All pipeline candidates were created from two proprietary platforms, ADAPTIRand ADAPTIR-FLEX. The Aptevo mission is to improve treatment outcomes and transform the lives of cancer patients. For more information, please visit www.aptevotherapeutics.com.
Safe Harbor Statement
This press release includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical fact, including, without limitation, Aptevo's expectations about the activity, efficacy, safety, tolerability and durability of its therapeutic candidates and potential use of any such candidates, including in combination with other drugs, as therapeutics for treatment of disease, its expectations regarding the effectiveness of its ADAPTIR and ADAPTIR-FLEX platforms, statements related to the progress of Aptevo's clinical programs, including statements related to anticipated clinical and regulatory milestones, whether further study of mipletamig in a Phase 1b dose optimization trial focusing on multiple doses of mipletamig in combination with venetoclax + azacitidine on a targeted patient population will continue to show remissions, whether further study of mipletamig in a Phase 1b dose-optimization trial will continue to report a favorable safety profile, let alone no instances of cytokine release syndrome, whether Aptevo's final trial results will vary from its earlier assessment, whether Aptevo's strategy will translate into an improved overall survival in AML, especially among patient subgroups with poor prognosis, whether further study of ALG.APV-527 across multiple tumor types will continue to show clinical benefit, the possibility and timing of interim data readouts for ALG.APV-527, development and continued development of Aptevo's current and potential future molecules, including its trispecific candidates, statements related to Aptevo's cash position and balance sheet, statements related to Aptevo's ability to generate stockholder value, whether Aptevo will continue to have momentum in its business in the future, statements regarding Aptevo's leadership transition, strategic direction, business outlook, expected future performance and any other statements containing the words "may," "continue to," "believes," "knows," "expects," "optimism," "potential," "designed," "promising," "plans," "will" and similar expressions are intended to identify forward-looking statements. These forward-looking statements are based on Aptevo's current intentions, beliefs, and expectations regarding future events. Aptevo cannot guarantee that any forward-looking statement will be accurate. Investors should realize that if underlying assumptions prove inaccurate or unknown risks or uncertainties materialize, actual results could differ materially from Aptevo's expectations. Investors are, therefore, cautioned not to place undue reliance on any forward-looking statement.
There are several important factors that could cause Aptevo's actual results to differ materially from those indicated by such forward-looking statements, including a deterioration in Aptevo's business or prospects; further assessment of preliminary or interim data or different results from later clinical trials; adverse events and unanticipated problems, adverse developments in clinical development, including unexpected safety issues observed during a clinical trial; and changes in regulatory, social, macroeconomic and political conditions. For instance, actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including the uncertainties inherent in the results of preliminary or interim data and preclinical studies being predictive of the results of later-stage clinical trials, initiation, enrollment and maintenance of patients, and the completion of clinical trials, the availability and timing of data from ongoing clinical trials, the trial design includes combination therapies that may make it difficult to accurately ascertain the benefits of mipletamig, expectations for the timing and steps required in the regulatory review process, expectations for regulatory approvals, the impact of competitive products, our ability to enter into agreements with strategic partners or raise funds on acceptable terms or at all and other matters that could affect the availability or commercial potential of Aptevo's product candidates, business or economic disruptions due to catastrophes or other events, including natural disasters or public health crises, geopolitical risks, including the current war between Russia and Ukraine, the United States and Iran and any other military event that could evolve out of any of the current conflicts, and macroeconomic conditions such as economic uncertainty, imposition of tariffs, rising inflation and interest rates, continued market volatility and decreased consumer confidence. These risks are not exhaustive, Aptevo faces known and unknown risks. Additional risks and factors that may affect results are set forth in Aptevo's filings with the Securities and Exchange Commission, including its Annual Report on Form 10-K for the fiscal year ended December 31, 2025, and its subsequent reports on Form 10-Q and current reports on Form 8-K. The foregoing sets forth many, but not all, of the factors that could cause actual results to differ from Aptevo's expectations in any forward-looking statement. Any forward-looking statement speaks only as of the date of this press release, and, except as required by law, Aptevo does not assume any obligation to update any forward-looking statement to reflect new information, events, or circumstances.
CONTACT:
Miriam Weber Miller
Vice President, Investor Relations & Corporate Communications
Aptevo Therapeutics
Email: [email protected] or [email protected]
Phone: 206-859-6628
SOURCE: Aptevo Therapeutics
View the original press release on ACCESS Newswire
R.Chavez--AT
