
-
 Blasts rock Kyiv in rare daytime Russian attack: AFP
Blasts rock Kyiv in rare daytime Russian attack: AFP
-
Israel announces Lebanon ground assault against Hezbollah

-
 AI to drive growth despite geopolitics, Taiwan's Foxconn says
AI to drive growth despite geopolitics, Taiwan's Foxconn says
-
Indonesia police seek four suspects for acid attack on activist

-
 Iranian protesters fled 'hell' at home, watch war from exile
Iranian protesters fled 'hell' at home, watch war from exile
-
'Rein in your heart': I.Coast women trapped by divorce taboo

-
 Five top moments from the Oscars
Five top moments from the Oscars
-
Filipinas seek abortions online in largely Catholic nation

-
 With Oscar race locked, actresses celebrate backstage anyway
With Oscar race locked, actresses celebrate backstage anyway
-
Chinese firms seek to loosen West's grip on lucrative snow business

-
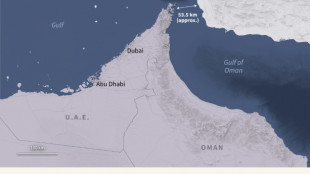 Japan not currently mulling maritime ops despite Trump pressure over Hormuz
Japan not currently mulling maritime ops despite Trump pressure over Hormuz
-
'One Battle After Another' wins best picture Oscar

-
 South Koreans bask in Oscars triumph for 'KPop Demon Hunters'
South Koreans bask in Oscars triumph for 'KPop Demon Hunters'
-
'One Battle After Another' dominates Oscars

-
 Oil hovers around $100, stocks mixed as Iran war rages
Oil hovers around $100, stocks mixed as Iran war rages
-
Future looks bright for Ireland and Farrell, says ex-Irish prop Ross

-
 Epic Six Nations raises defensive questions before Rugby World Cup
Epic Six Nations raises defensive questions before Rugby World Cup
-
Ryan Coogler: from indie to blockbuster to Oscar

-
 English sides seek redemption from Champions League reality check
English sides seek redemption from Champions League reality check
-
Jessie Buckley: From reality TV hopeful to Oscar winner

-
 Paul Thomas Anderson: eclectic filmmaker, critical darling
Paul Thomas Anderson: eclectic filmmaker, critical darling
-
Michael B. Jordan battles his way to Oscar for 'Sinners'

-
 20 Thai sailors return home after vessel attacked in Gulf
20 Thai sailors return home after vessel attacked in Gulf
-
Norway's Oscar winner 'Sentimental Value': a failing father seeks redemption

-
 Races off, Mercedes dominance, McLaren woe: China GP talking points
Races off, Mercedes dominance, McLaren woe: China GP talking points
-
Indonesia firms in palm oil fraud probe supplied fuel majors

-
 Sean Penn: Hollywood's rebel with a cause wins third Oscar
Sean Penn: Hollywood's rebel with a cause wins third Oscar
-
It's 'Sinners' vs 'One Battle' as the Oscars begin

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - March 16
InterContinental Hotels Group PLC Announces Transaction in Own Shares - March 16
-
GEN Receives Ethical Approval to Launch Phase II Clinical Trial for SUL-238 Targeting Mitochondrial Dysfunction in Patients with Parkinson’s Disease

-
 Sinner tops Medvedev to win first Indian Wells title
Sinner tops Medvedev to win first Indian Wells title
-
'KPop Demon Hunters' wins Oscar for best animated feature

-
 Left leads in Paris, far right eyes gains in France local polls: projections
Left leads in Paris, far right eyes gains in France local polls: projections
-
Amy Madigan wins Oscar as 'Weapons' villain Aunt Gladys

-
 Stars bring glamour to Oscars red carpet
Stars bring glamour to Oscars red carpet
-
Israel launches fresh strike on south Beirut

-
 Laporta reelected Barca president for next five years
Laporta reelected Barca president for next five years
-
Young outduels Fitzpatrick to claim Players victory

-
 AC Milan slip at Lazio as Como eye Champions League
AC Milan slip at Lazio as Como eye Champions League
-
Milan-Cortina Paralympics end as a 'beacon of unity'

-
 It's 'Sinners' vs 'One Battle' as Oscars day arrives
It's 'Sinners' vs 'One Battle' as Oscars day arrives
-
Oscars night: latest developments

-
 Sabalenka edges Rybakina to claim long-awaited Indian Wells title
Sabalenka edges Rybakina to claim long-awaited Indian Wells title
-
Myanmar's post-coup parliament sits packed with junta allies

-
 Stuttgart down Leipzig to strike blow in top-four race
Stuttgart down Leipzig to strike blow in top-four race
-
Gilgeous-Alexander keeps streak alive as Thunder down Wolves

-
 Tudor sees Tottenham 'turning point' in Liverpool draw
Tudor sees Tottenham 'turning point' in Liverpool draw
-
Espressos and red wine: Italy's baseball team captivates Classic

-
 Troubled Spurs snatch late draw at Liverpool, Man Utd boost top-four bid
Troubled Spurs snatch late draw at Liverpool, Man Utd boost top-four bid
-
USA win fifth straight gold in Paralympics ice hockey


GEN Receives Ethical Approval to Launch Phase II Clinical Trial for SUL-238 Targeting Mitochondrial Dysfunction in Patients with Parkinson’s Disease
GEN announces approval in the Netherlands to initiate patient enrollment in April 2026 for the Phase II Proof of Concept trial of its novel drug candidate SUL-238, Targeting Mitochondrial Dysfunction in Patients with Parkinson's Disease.
ANKARA, TR / ACCESS Newswire / March 16, 2026 / GEN Pharmaceuticals (GENIL.IS), Türkiye's leading specialty pharmaceutical company, announced today that it has received approval in the Netherlands from the BEBO Foundation for the Assessment of Ethics of Biomedical Research (Stichting Beoordeling Ethiek Biomedisch Onderzoek) to proceed with Phase II Proof of Concept Trial for SUL-238 Targeting Mitochondrial Dysfunction in Patients with Parkinson's Disease. With this approval, GEN can now proceed to activate the clinical site in Groningen, the Netherlands. Patient enrollment is planned to start in April 2026.
Details of the Phase II trial, entitled "A Phase 2, Randomized, Double-blind, Placebo-Controlled, Single-Center Study to Assess the effects of SUL-238 on High Energy Phosphates with Magnetic Resonance Spectroscopy (31P-MRS) in patients with Early, untreated Parkinson's Disease ("SHEPHERD" STUDY)" can be viewed at clinicaltrials.gov.
Abidin Gülmüş, Chairman of GEN, stated:
"Following the ethical approval, we look forward to starting patient enrollment in the Netherlands.This Phase 2 trial will be another key milestone toward addressing neurodegenerative diseases at its biological foundation."
About Parkinson's Disease
Parkinson's Disease (PD) is the most common neurodegenerative movement disorder affecting up to 2% of those aged above 60 years. The disease follows a progressive course, and there are still no proven strategies for slowing the progression of PD. Mitochondrial dysfunction and oxidative damage are implicated in the pathophysiology of neurodegenerative disorders, including PD and Alzheimer's disease. Since mitochondria are highly multifunctional organelles, their integrity is essential for neuronal function and survival.
About SUL-238
SUL-238 is a novel, first-in-class, hibernation-inspired small molecule designed to target mitochondria, the 'powerhouse' of the cell. SUL-238 supports mitochondrial bioenergetics via complex I/IV activation and enhances mitochondrial function in various preclinical models for neurodegenerative, cardiovascular, and renal diseases, and also in accelerated aging. SUL-238 exhibits a favourable pharmacokinetic profile and a high cerebrospinal fluid penetration, and has undergone extensive safety evaluation in preclinical and clinical Phase 1 studies. GEN licenses SUL-238 from Sulfateq B.V. for neurodegenerative disease applications.
About GEN:
Founded in 1998, GEN is Türkiye's leading specialty pharmaceutical company, focused on developing innovative therapies across multiple therapeutic areas. Through significant R&D investments and global collaborations, GEN is committed to advancing healthcare worldwide. The company develops and manufactures high-quality, competitive products at its GMP-certified production facility and continues its bold efforts in original drug development via two dedicated R&D centers.
About Sulfateq:
Sulfateq B.V. is a Dutch biotech company in Groningen (the Netherlands) that fosters strategic collaborations with academic and industrial research centres to accelerate the development of innovative new medicines. It has developed a novel class of small-molecule SUL compounds that maintain mitochondrial health.
For more information:
www.genilac.com.tr
www.sulfateqbv.com
Press Contacts:
Bulutay GÜNEŞ, Sr. Head of Corporate Brand
[email protected]
Fatih GÖREN, Investor Relations Manager
[email protected]
Kees van der Graaf, Chief Executive Officer
[email protected]
SOURCE: GEN Ilac ve Saglik Urunleri A.S.
View the original press release on ACCESS Newswire
K.Hill--AT


