
-
 Oil prices tumble, stocks rally on Mideast peace hopes
Oil prices tumble, stocks rally on Mideast peace hopes
-
Maybach: Between Glory and a Turning Point

-
 German business morale falls as war puts recovery on ice: survey
German business morale falls as war puts recovery on ice: survey
-
Labubu maker Pop Mart's shares fall 23% despite surging earnings

-
 ECB won't be 'paralysed' in face of energy shock: Lagarde
ECB won't be 'paralysed' in face of energy shock: Lagarde
-
Iran hits targets across Middle East after Trump signals talks progress

-
 McEvoy says best is to come after breaking long-standing swim record
McEvoy says best is to come after breaking long-standing swim record
-
Goat vs gecko: A tiny Caribbean island faces wildlife showdown

-
 Japan PM asks IEA chief to prepare additional 'coordinated release' of oil
Japan PM asks IEA chief to prepare additional 'coordinated release' of oil
-
Hungary's hard-pressed LGBTQ people say Orban exit is only half battle

-
 Belarus leader visits North Korea for first time
Belarus leader visits North Korea for first time
-
'No heavier burden': the decades-long search for Kosovo war missing

-
 Exotic pet trade thrives in China despite welfare concerns
Exotic pet trade thrives in China despite welfare concerns
-
Iran fires missile salvo after Trump signals progress in talks

-
 BTS concert drew 18.4 million viewers, says Netflix
BTS concert drew 18.4 million viewers, says Netflix
-
OSCE's 'chaotic' Ukraine evacuation put staff at risk: leaked report

-
 Top WTO official sounds fertiliser warning over Middle East war
Top WTO official sounds fertiliser warning over Middle East war
-
France and Brazil weigh up World Cup prospects in glamour friendly

-
 Italy hoping to end World Cup pain as play-offs loom
Italy hoping to end World Cup pain as play-offs loom
-
Dirty diapers born again in Japan recycling breakthrough

-
 Verstappen's Japan GP win streak under threat as Mercedes dominate
Verstappen's Japan GP win streak under threat as Mercedes dominate
-
Crude tumbles, stocks rally on hopes for Iran war de-escalation

-
 Gauff outlasts Bencic to reach Miami semi-finals
Gauff outlasts Bencic to reach Miami semi-finals
-
'Hero' Australian dog who saved 100 koalas retires

-
 Underdogs chase World Cup berths in Mexico playoff tournament
Underdogs chase World Cup berths in Mexico playoff tournament
-
Pope heads to tiny Catholic Monaco

-
 Meet the four astronauts set to voyage around the Moon
Meet the four astronauts set to voyage around the Moon
-
Artemis 2 Moon mission: a primer

-
 It's go time: historic Moon mission set for lift-off
It's go time: historic Moon mission set for lift-off
-
Denmark's PM Mette Frederiksen, tenacious and tough on migration

-
 OpenAI kills Sora video app in pivot toward business tools
OpenAI kills Sora video app in pivot toward business tools
-
Danish PM's left-wing bloc wins election, but no majority

-
 Gold Basin Announces Termination of Charles Straw for Cause
Gold Basin Announces Termination of Charles Straw for Cause
-
ParkerVision Completes Appeal Briefing

-
 Bridgeline's HawkSearch and Znode Partner to Bring AI-Powered Search & Product Discovery to Enterprise B2B eCommerce
Bridgeline's HawkSearch and Znode Partner to Bring AI-Powered Search & Product Discovery to Enterprise B2B eCommerce
-
Cheech and Chong's Global Holding Company Partners with Breakthru Beverage Group to Expand Hemp THC Beverage Distribution

-
 Zedge Increases Quarterly Cash Dividend by 25%
Zedge Increases Quarterly Cash Dividend by 25%
-
Who Does the Best Breast Augmentation in Vancouver, BC?

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - March 25
InterContinental Hotels Group PLC Announces Transaction in Own Shares - March 25
-
Apex Critical Metals Appoints Zayn Kalyan to Board of Directors

-
 Troy Mick Appointed Head of SSS Phoenix Academy at Fire 'n' Ice Arena
Troy Mick Appointed Head of SSS Phoenix Academy at Fire 'n' Ice Arena
-
Brazil court grants house arrest for jailed Bolsonaro

-
 Sinner downs Michelsen to reach Miami Open quarter-finals
Sinner downs Michelsen to reach Miami Open quarter-finals
-
Advantage Arsenal in women's Champions League quarter-final against Chelsea

-
 Garner dreams of World Cup glory in bid to replicate England under-21 success
Garner dreams of World Cup glory in bid to replicate England under-21 success
-
New Mexico jury finds Meta liable for endangering children

-
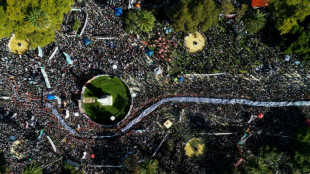 Huge crowd in Buenos Aires marks 50 years since Argentina's coup
Huge crowd in Buenos Aires marks 50 years since Argentina's coup
-
Oil, stock trading spiked before Trump's Iran remarks

-
 Colombia military plane crash death toll rises to 69
Colombia military plane crash death toll rises to 69
-
Trump adds Columbus statue, walkway in latest White House makeover


Revelation Biosciences, Inc. Announces Financial Results For the Three and Twelve Months Ended December 31, 2025
SAN DIEGO, CA / ACCESS Newswire / February 26, 2026 / Revelation Biosciences, Inc. (NASDAQ:REVB) (the "Company" or "Revelation"), a clinical-stage life sciences company focused on rebalancing inflammation to optimize health, today reported its financial results for the three and twelve months ended December 31, 2025.
Corporate Highlights
Announced Positive Results from PRIME Clinical Study in late-stage chronic kidney disease patients
Announced agreement with FDA on a single adaptive Phase 2/3 study for approval of Gemini for treatment of acute kidney injury
Received net proceeds of $6.7 million from warrant inducement in January 2026
Clinical data on the potential therapeutic benefit of Gemini for the treatment of acute kidney injury (AKI) and chronic kidney disease (CKD) will be presented at the International Conference on Advances in Critical Care Nephrology in San Diego (March 29 - April 1, 2026)
"2025 was a positive year for Revelation with significant advancement of the Gemini program," said James Rolke, Chief Executive Officer of Revelation. "We look forward to building on this momentum in 2026 to expeditiously bring Gemini to patients in need and adding to shareholder value."
Results of Operations
As of December 31, 2025, Revelation had $10.7 million in cash and cash equivalents, compared to $6.5 million as of December 31, 2024. The increase in cash and cash equivalents was primarily due to net cash proceeds from the May 2025 public offering and the September 2025 warrant inducement, offset by cash used for operating activities. Based on current operating plans and projections, Revelation believes its current cash and cash equivalents are sufficient to fund operations into the first quarter of 2027.
Net cash used for operating activities for the twelve months ended December 31, 2025 was $8.3 million compared to net cash used for operating activities of $18.3 million for the same period in 2024. Net loss for the three months ended December 31, 2025 was $2.5 million, or $(1.65) basic and diluted net loss per share, compared to a net loss of $1.7 million, or $(59.76) basic and diluted net loss per share for the same period in 2024. Net loss for the year ended December 31, 2025 was $8.9 million, or $(23.95) basic and diluted net loss per share compared to net loss of $15.0 million, or $(1,052.16) basic and diluted net loss per share for the year ended December 31, 2024.
About Gemini
Gemini is the Company's proprietary formulation of phosphorylated hexaacyl disaccharide (PHAD®), a toll-like receptor 4 (TLR4) agonist. TLR4 stimulation with Gemini rebalances the innate immune response and has been demonstrated to have the potential to treat acute and chronic diseases associated with dysregulated inflammation. Gemini is currently being evaluated as a potential treatment for acute kidney injury (GEM-AKI); Gemini is also being developed as a treatment for chronic kidney disease (GEM-CKD), as a treatment to reduce hyperinflammation and infection associated with severe burn (GEM-PBI), and as a treatment to prevent post-surgical infection (GEM-PSI). The potential of Gemini to correct dysregulated inflammation has been demonstrated in multiple preclinical models of AKI, CKD, and infection, as well as in two phase 1 clinical studies. See additional detail here.
About Revelation Biosciences, Inc.
Revelation Biosciences, Inc. is a clinical stage life sciences company focused on rebalancing inflammation using its proprietary formulation, Gemini. Revelation has multiple ongoing programs to evaluate Gemini as a treatment for acute kidney injury, a treatment of chronic kidney disease, prevention of post-surgical infection, and a treatment to reduce hyperinflammation and infection associated with severe burn.
For more information, please visit www.RevBiosciences.com.
Forward-Looking Statements
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These forward-looking statements are generally identified by the words "anticipate", "believe", "expect", "estimate", "plan", "outlook", and "project" and other similar expressions. We caution investors that forward-looking statements are based on management's expectations and are only predictions or statements of current expectations and involve known and unknown risks, uncertainties and other factors that may cause actual results to be materially different from those anticipated by the forward-looking statements. Revelation cautions readers not to place undue reliance on any such forward looking statements, which speak only as of the date they were made. The following factors, among others, could cause actual results to differ materially from those described in these forward-looking statements: the ability of Revelation to meet its financial and strategic goals, due to, among other things, competition; the ability of Revelation to grow and manage growth profitability and retain its key employees; the possibility that the Revelation may be adversely affected by other economic, business, and/or competitive factors; risks relating to the successful development of Revelation's product candidates; the ability to successfully complete planned clinical studies of its product candidates; the risk that we may not fully enroll our clinical studies or enrollment will take longer than expected; risks relating to the occurrence of adverse safety events and/or unexpected concerns that may arise from data or analysis from our clinical studies; changes in applicable laws or regulations; expected initiation of the clinical studies, the timing of clinical data; the outcome of the clinical data, including whether the results of such study is positive or whether it can be replicated; the outcome of data collected, including whether the results of such data and/or correlation can be replicated; the timing, costs, conduct and outcome of our other clinical studies; the anticipated treatment of future clinical data by the FDA, the EMA or other regulatory authorities, including whether such data will be sufficient for approval; the success of future development activities for its product candidates; potential indications for which product candidates may be developed; the ability of Revelation to maintain the listing of its securities on NASDAQ; the expected duration over which Revelation's balances will fund its operations; and other risks and uncertainties described herein, as well as those risks and uncertainties discussed from time to time in other reports and other public filings with the SEC by Revelation.
REVELATION BIOSCIENCES, INC.
Consolidated Statements of Operations
Three Months Ended | Year Ended | |||||||||||
2025 | 2024 | 2025 | 2024 | |||||||||
Operating expenses: | ||||||||||||
Research and development | $ | 964,189 | $ | 605,504 | $ | 4,063,857 | $ | 3,548,996 | ||||
General and administrative | 1,607,398 | 1,148,384 | 5,006,957 | 4,426,113 | ||||||||
Total operating expenses | 2,571,587 | 1,753,888 | 9,070,814 | 7,975,109 | ||||||||
Loss from operations | (2,571,587 | ) | (1,753,888 | ) | (9,070,814 | ) | (7,975,109 | ) | ||||
Other income (expense): | ||||||||||||
Change in fair value of warrant liability | 87 | 2,557 | 2,158 | 81,441 | ||||||||
Other income (expense), net | 60,493 | 25,612 | 155,007 | (7,144,868 | ) | |||||||
Total other income (expense), net | 60,580 | 28,169 | 157,165 | (7,063,427 | ) | |||||||
Net loss | $ | (2,511,007 | ) | $ | (1,725,719 | ) | $ | (8,913,649 | ) | $ | (15,038,536 | ) |
Deemed dividends | - | - | (5,951,528 | ) | - | |||||||
Net loss attributable to common stockholders | (2,511,007 | ) | (1,725,719 | ) | (14,865,177 | ) | (15,038,536 | ) | ||||
Net loss per share, basic and diluted | $ | (1.65 | ) | $ | (59.76 | ) | $ | (23.95 | ) | $ | (1,052.16 | ) |
Weighted-average shares used to compute net loss per share, basic and diluted | 1,524,011 | 28,876 | 620,785 | 14,293 | ||||||||
REVELATION BIOSCIENCES, INC.
Consolidated Balance Sheets
December 31, | December 31, | |||
ASSETS | ||||
Current assets: | ||||
Cash and cash equivalents | $ | 10,700,331 | $ | 6,499,018 |
Prepaid expenses and other current assets | 111,297 | 66,699 | ||
Total current assets | 10,811,628 | 6,565,717 | ||
Property and equipment, net | 18,067 | 56,332 | ||
Operating lease right-of-use asset | 722,288 | - | ||
Other assets | 30,941 | - | ||
Total assets | $ | 11,582,924 | $ | 6,622,049 |
LIABILITIES AND STOCKHOLDERS' EQUITY | ||||
Current liabilities: | ||||
Accounts payable | $ | 577,501 | $ | 783,621 |
Accrued expenses | 1,397,644 | 1,130,046 | ||
Operating lease liability | 23,013 | - | ||
Total current liabilities | 1,998,158 | 1,913,667 | ||
Operating lease liability, net of current portion | 723,771 | - | ||
Total liabilities | 2,721,929 | 1,913,667 | ||
Commitments and Contingencies (Note 4) | ||||
Stockholders' equity: | ||||
Common Stock, $0.001 par value; 500,000,000 shares authorized at December 31, 2025 and December 31, 2024 and 1,583,969 and 43,526 issued and outstanding at December 31, 2025 and December 31, 2024, respectively | 1,584 | 44 | ||
Additional paid-in-capital | 58,278,698 | 45,213,976 | ||
Accumulated deficit | (49,419,287 | ) | (40,505,638 | ) |
Total stockholders' equity | 8,860,995 | 4,708,382 | ||
Total liabilities and stockholders' equity | $ | 11,582,924 | $ | 6,622,049 |
Company Contacts
Mike Porter
Investor Relations
Porter LeVay & Rose Inc.
Email: [email protected]
Chester Zygmont, III
Chief Financial Officer
Revelation Biosciences Inc.
Email: [email protected]
SOURCE: Revelation Biosciences, Inc.
View the original press release on ACCESS Newswire
A.Moore--AT
