
-
 UBS first-quarter profits jump 80% on investment banking
UBS first-quarter profits jump 80% on investment banking
-
France's 'roadmap' to exit fossil fuels by 2050

-
 Chelsea captain Millie Bright retires
Chelsea captain Millie Bright retires
-
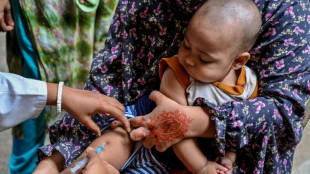
Bangladesh measles outbreak kills over 220 children since March
-
 Finnish lift maker Kone acquires German rival TKE, creating giant
Finnish lift maker Kone acquires German rival TKE, creating giant
-
Hungary's Magyar visits Brussels seeking to unblock EU billions

-
 Diving robot explores mystery of France's deepest shipwreck
Diving robot explores mystery of France's deepest shipwreck
-
Thai ex-PM Thaksin to be released from prison next month

-
 Welsh rugby great North to hang up his boots
Welsh rugby great North to hang up his boots
-
Much-needed rains revive Iraq's fabled Mesopotamian Marshes

-
 French teen in straw licking case allowed to leave Singapore
French teen in straw licking case allowed to leave Singapore
-
EU chief says Kremlin imposing 'digital Iron Curtain' on Russians

-
 South Korean court hikes ex-president's sentence for obstructing justice
South Korean court hikes ex-president's sentence for obstructing justice
-
Adidas reports higher profits but warns of 'volatile' climate

-
 TotalEnergies first-quarter profits surge amid Middle East war
TotalEnergies first-quarter profits surge amid Middle East war
-
Sri Lanka government 'temporarily' takes over cricket board

-
 EU finds Meta failing to keep under-13s off Facebook, Instagram
EU finds Meta failing to keep under-13s off Facebook, Instagram
-
King Charles to stress UK-US cultural, trade ties in New York

-
 US judge orders Purdue Pharma to pay billions ahead of bankruptcy
US judge orders Purdue Pharma to pay billions ahead of bankruptcy
-
'Jurassic Park' star Sam Neill says cancer-free after gene therapy

-
 US opioid crisis victims testify at emotional Purdue Pharma hearing
US opioid crisis victims testify at emotional Purdue Pharma hearing
-
Australian climber on record sea-to-summit Everest bid

-
 Indian opposition slams Nicobar megaport plan as 'destruction'
Indian opposition slams Nicobar megaport plan as 'destruction'
-
Pentagon chief to testify on Iran war, peace efforts stall

-
 Anxiety, resentment around AI spur violence against tech's figureheads
Anxiety, resentment around AI spur violence against tech's figureheads
-
Mercedes-Benz profit slides amid cutthroat Chinese market

-
 Hungary's Magyar to push post-Orban EU reset on Brussels visit
Hungary's Magyar to push post-Orban EU reset on Brussels visit
-
Going online helps Pakistan's women doctors back to work

-
 Wembanyama's Spurs advance in NBA playoffs, 76ers stay alive
Wembanyama's Spurs advance in NBA playoffs, 76ers stay alive
-
Tropical forest loss eases after record year: researchers

-
 Tigres edges Nashville in CONCACAF Champions Cup first leg
Tigres edges Nashville in CONCACAF Champions Cup first leg
-
New Zealand officials reject statue remembering Japan's sex slaves

-
 King Charles, Trump toast ties despite Iran tensions
King Charles, Trump toast ties despite Iran tensions
-
Japan cleaner goes viral with spa-like service for plushies

-
 What we learned from cycling's Spring Classics
What we learned from cycling's Spring Classics
-
Villa, Forest revive European glory days in semi-final showdown

-
 Remarkable, ramshackle Rayo chasing Conference League dream amid chaos
Remarkable, ramshackle Rayo chasing Conference League dream amid chaos
-
Unbeaten records on the line for Inoue-Nakatani superfight in Tokyo

-
 Cheaper, cleaner electric trucks overhaul China's logistics
Cheaper, cleaner electric trucks overhaul China's logistics
-
Stocks swing, oil edges up with Iran war peace talks stalled

-
 Europe climate report signals rising extremes
Europe climate report signals rising extremes
-
Sexual violence in Sudan triggers mental health crisis: UN

-
 The loyal, lonely keepers of Sudan's pyramids
The loyal, lonely keepers of Sudan's pyramids
-
'Final mission': NZ name star trio for T20 World Cup defence

-
 Embiid-led 76ers beat Boston to avoid NBA playoff exit
Embiid-led 76ers beat Boston to avoid NBA playoff exit
-
An experimental cafe run by AI opens in Stockholm

-
 Exiting fossil fuels key to energy security: nations at Colombia talks
Exiting fossil fuels key to energy security: nations at Colombia talks
-
Jerome Powell: Fed chair who stood up to Trump set to finish tenure on top

-
 All eyes on Powell with US Fed expected to hold rates steady
All eyes on Powell with US Fed expected to hold rates steady
-
Pentagon makes deal to expand use of Google AI: reports


Onco-Innovations Commences Activities to Manufacture Nanoparticle Formulation with Dalton Pharma to Supply Preclinical Animal Studies
VANCOUVER, BC / ACCESS Newswire / September 19, 2025 / Onco-Innovations Limited (CBOE CA:ONCO )(OTCQB:ONNVF )(Frankfurt:W1H) (WKN: A3EKSZ ) (" Onco " or the " Company ") is pleased to announce that Dalton Pharma Services ("Dalton") has commenced activities to manufacture Onco's proprietary nanoparticle formulation, also know as PEO-b-PBz-CL 1 , a key component of the Company's novel drug delivery platform which is designed for use in conjunction with Onco's proprietary Polynucleotide Kinase Phosphatase (PNKP) inhibitor. The manufacturing initiative is intended to generate the nanoparticle material required to advance preclinical studies.
The production campaign will include process optimization and scale-up activities to establish reproducible methods for the synthesis of PEO-b-PBz-CL, while ensuring that the formulation meets quality specifications for purity, molecular weight, and residual solvent levels. Dalton has already demonstrated initial feasibility at the 10 grams scale and will now focus on reducing residual catalyst content and scaling production to 100 grams, followed by a demonstration batch at the 350 grams scale. These activities are designed to provide sufficient quantities of nanoparticle material for formulation development, while also generating the analytical data necessary to support regulatory filings and the eventual transition to Good Manufacturing Practice (GMP) manufacturing.
By engaging Dalton, Onco benefits from the technical expertise and regulatory experience of one of Canada's leading contract development and manufacturing organizations. Dalton's work will provide a comprehensive development package, including full analytical characterization and process documentation, to ensure a smooth path toward clinical readiness of the nanoparticle-formulated platform.
" Advancing the nanoparticle formulation demonstrates will help inform our next phase of development, advancing our pathway toward the clinical opportunities we are determined to reach, " stated Thomas O'Shaughnessy, CEO of Onco-Innovations.
About Onco-Innovations Limited
Onco-Innovations is a Canadian-based company dedicated to cancer research and treatment, specializing in oncology. Onco's mission is to pursue the prevention and treatment of cancer through pioneering research and innovative solutions. The company has secured an exclusive worldwide license to patented technology that targets solid tumours.
ON BEHALF OF ONCO-INNOVATIONS LIMITED,
" Thomas O'Shaughnessy "
Chief Executive Officer
For more information, please contact:
Thomas O'Shaughnessy
Chief Executive Officer
Tel: + 1 888 261 8055
[email protected]
Forward-Looking Statements Caution. This news release contains forward-looking statements, including in relation to the benefits expected to be realized under the agreement with Dalton, and the Company's ability to move forward with its plans for manufacturing and further testing and development of its Technologies, the prospects of the Company, and the Company's business and plans generally, and other statements that are not historical facts. Forward-looking statements are often identified by terms such as "will", "may", "potential", "should", "anticipate", "expects" and similar expressions. All statements other than statements of historical fact, included in this release are forward-looking statements that involve risks and uncertainties. There can be no assurance that such statements will prove to be accurate and actual results and future events could differ materially from those anticipated in such statements. The reader is cautioned that assumptions used in the preparation of any forward-looking information may prove to be incorrect. Events or circumstances may cause actual results to differ materially from those predicted, as a result of numerous known and unknown risks, uncertainties, and other factors, many of which are beyond the control of the Company. The reader is cautioned not to place undue reliance on any forward-looking information. Such information, although considered reasonable by management at the time of preparation, may prove to be incorrect and actual results may differ materially from those anticipated. Forward-looking statements contained in this news release are expressly qualified by this cautionary statement. The forward-looking statements contained in this news release are made as of the date of this news release and the Company will update or revise publicly any of the included forward-looking statements as expressly required by applicable law.
1poly(ethylene oxide)-block-poly(α-benzyl carboxylate ε-caprolactone)
SOURCE: Onco-Innovations LimitedView the original press release on ACCESS Newswire
O.Brown--AT
