
-
 Emery aims to write 'new chapter' in Europe with Villa
Emery aims to write 'new chapter' in Europe with Villa
-
US Supreme Court curbs race-based voting maps in landmark ruling

-
 Guerrillas claim deadly Colombia attack, say it was an 'error'
Guerrillas claim deadly Colombia attack, say it was an 'error'
-
Trump warns Iran better 'get smart soon' and accept nuclear deal

-
 UN experts urge Saudi labour practices switch before World Cup
UN experts urge Saudi labour practices switch before World Cup
-
Oil spikes while stocks slide ahead of US Fed rate decision

-
 US Fed chief's plans in focus as central bank set to hold rates steady
US Fed chief's plans in focus as central bank set to hold rates steady
-
Tuareg rebels vow Mali junta 'will fall', north will be captured

-
 German inflation jumps in April as energy costs surge
German inflation jumps in April as energy costs surge
-
Was PSG against Bayern the Champions League's greatest ever game?

-
 UBS first-quarter profits jump 80% on investment banking
UBS first-quarter profits jump 80% on investment banking
-
France's 'roadmap' to exit fossil fuels by 2050

-
 Chelsea captain Millie Bright retires
Chelsea captain Millie Bright retires
-
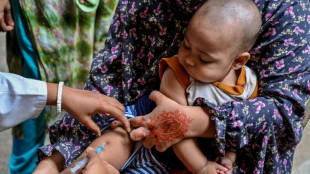
Bangladesh measles outbreak kills over 220 children since March
-
 Finnish lift maker Kone acquires German rival TKE, creating giant
Finnish lift maker Kone acquires German rival TKE, creating giant
-
Hungary's Magyar visits Brussels seeking to unblock EU billions

-
 Diving robot explores mystery of France's deepest shipwreck
Diving robot explores mystery of France's deepest shipwreck
-
Thai ex-PM Thaksin to be released from prison next month

-
 Welsh rugby great North to hang up his boots
Welsh rugby great North to hang up his boots
-
Much-needed rains revive Iraq's fabled Mesopotamian Marshes

-
 French teen in straw licking case allowed to leave Singapore
French teen in straw licking case allowed to leave Singapore
-
EU chief says Kremlin imposing 'digital Iron Curtain' on Russians

-
 South Korean court hikes ex-president's sentence for obstructing justice
South Korean court hikes ex-president's sentence for obstructing justice
-
Adidas reports higher profits but warns of 'volatile' climate

-
 TotalEnergies first-quarter profits surge amid Middle East war
TotalEnergies first-quarter profits surge amid Middle East war
-
Sri Lanka government 'temporarily' takes over cricket board

-
 EU finds Meta failing to keep under-13s off Facebook, Instagram
EU finds Meta failing to keep under-13s off Facebook, Instagram
-
King Charles to stress UK-US cultural, trade ties in New York

-
 US judge orders Purdue Pharma to pay billions ahead of bankruptcy
US judge orders Purdue Pharma to pay billions ahead of bankruptcy
-
'Jurassic Park' star Sam Neill says cancer-free after gene therapy

-
 US opioid crisis victims testify at emotional Purdue Pharma hearing
US opioid crisis victims testify at emotional Purdue Pharma hearing
-
Australian climber on record sea-to-summit Everest bid

-
 Indian opposition slams Nicobar megaport plan as 'destruction'
Indian opposition slams Nicobar megaport plan as 'destruction'
-
Pentagon chief to testify on Iran war, peace efforts stall

-
 Anxiety, resentment around AI spur violence against tech's figureheads
Anxiety, resentment around AI spur violence against tech's figureheads
-
Mercedes-Benz profit slides amid cutthroat Chinese market

-
 Hungary's Magyar to push post-Orban EU reset on Brussels visit
Hungary's Magyar to push post-Orban EU reset on Brussels visit
-
Going online helps Pakistan's women doctors back to work

-
 Wembanyama's Spurs advance in NBA playoffs, 76ers stay alive
Wembanyama's Spurs advance in NBA playoffs, 76ers stay alive
-
Tropical forest loss eases after record year: researchers

-
 Tigres edges Nashville in CONCACAF Champions Cup first leg
Tigres edges Nashville in CONCACAF Champions Cup first leg
-
New Zealand officials reject statue remembering Japan's sex slaves

-
 King Charles, Trump toast ties despite Iran tensions
King Charles, Trump toast ties despite Iran tensions
-
Japan cleaner goes viral with spa-like service for plushies

-
 What we learned from cycling's Spring Classics
What we learned from cycling's Spring Classics
-
Villa, Forest revive European glory days in semi-final showdown

-
 Remarkable, ramshackle Rayo chasing Conference League dream amid chaos
Remarkable, ramshackle Rayo chasing Conference League dream amid chaos
-
Unbeaten records on the line for Inoue-Nakatani superfight in Tokyo

-
 Cheaper, cleaner electric trucks overhaul China's logistics
Cheaper, cleaner electric trucks overhaul China's logistics
-
Stocks swing, oil edges up with Iran war peace talks stalled


Dermata Therapeutics Announces Presentation of Abstract at the European Academy of Dermatology and Venereology Congress 2025
- Abstract highlights additional primary and secondary data from Phase 3 STAR-1 clinical study of XYNGARI™ for the treatment of moderate-to-severe acne -
- Abstract highlights additional primary and secondary data from Phase 3 STAR-1 clinical study of XYNGARI™ for the treatment of moderate-to-severe acne -
SAN DIEGO, CA / ACCESS Newswire / September 17, 2025 / Dermata Therapeutics, Inc. (Nasdaq:DRMA)(Nasdaq:DRMAW) ("Dermata" or the "Company"), a science-driven leader in dermatologic solutions, today announced the presentation of an abstract from its XYNGARI™ (also known as DMT310) Phase 3 Spongilla Treatment of Acne Research clinical trial (STAR-1) will be presented at the European Academy of Dermatology and Venereology (EADV) Congress 2025 being held in Paris, France, from September 17-20th.
Details of the abstract are as follows:
Abstract Title: Once Weekly Topical Treatment with DMT310 Demonstrates Significant and Early Onset of Effect in Patients with Moderate to Severe Acne Vulgaris - Results from the STAR-1 Phase 3 Study
Abstract Number: P3243
The full abstract will be released on Tuesday, September 16, 2025, at 10:00pm (PST). For more information about the EADV Congress 2025, please visit https://eadv.org/congress/
Click here to view the EADV abstract.
Forward-Looking Statements
Statements in this press release that are not strictly historical in nature are forward-looking statements. These statements are based on the Company's current beliefs and expectations and new risks may emerge from time to time. Forward-looking statements are subject to known and unknown risks, uncertainties, assumptions and other factors including, but are not limited to, statements related to: Dermata's shift to prioritize OTC dermatology products; the anticipated benefits of the strategic shift; the anticipated benefits of Dermata's strategic shift, including acceleration of its path to commercialization, reduction of regulatory burdens, and expansion into broader consumer markets; the expected timing and success of any planned or future OTC product launches; risks that clinical trials may not be predictive of real-world results or of results of subsequent clinical trials; risks that current clinical trials will lead to further product development by the Company; and other factors described in the Company's filings with the Securities and Exchange Commission. These forward-looking statements are generally identified by the use of such words as "may," "could," "should," "would," "believe," "anticipate," "forecast," "estimate," "expect," "intend," "plan," "continue," "outlook," "will," "potential" and similar statements of a future or forward-looking nature. These statements are only predictions based on current information and expectations and involve a number of risks and uncertainties. Actual events or results may differ materially from those projected in any of such statements due to various factors, including the risks and uncertainties inherent in drug development, approval and commercialization, and the fact that past results of clinical trials may not be indicative of future trial results. For a discussion of these and other factors, please refer to Dermata's filings with the Securities and Exchange Commission. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. This caution is made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. All forward-looking statements are qualified in their entirety by this cautionary statement and Dermata undertakes no obligation to revise or update this press release to reflect events or circumstances after the date hereof, except as required by law.
Dermata Investors:
Cliff Mastricola
Investor Relations
[email protected]
SOURCE: Dermata Therapeutics
View the original press release on ACCESS Newswire
N.Mitchell--AT
