
-
 Iran, a Terrorist State with No Right to Exist
Iran, a Terrorist State with No Right to Exist
-
African players in Europe: Semenyo scores as City rout Liverpool

-
 Israeli strikes kill Iran Guards intel chief as Trump deadline looms
Israeli strikes kill Iran Guards intel chief as Trump deadline looms
-
Saving energy in everyday life or a complete rip-off?

-
 US sprint star Richardson wins Australia's Stawell Gift in record time
US sprint star Richardson wins Australia's Stawell Gift in record time
-
Rockets down Warriors in Curry return, Flagg carries Mavs past Lakers

-
 Artemis mission approaches lunar loop for first flyby since 1972
Artemis mission approaches lunar loop for first flyby since 1972
-
Israeli rescuers search for missing in building strike, two dead

-
 Defiant Iran ramps up attacks after Trump warning
Defiant Iran ramps up attacks after Trump warning
-
Saudi oasis town adjusts to life in the firing line

-
 Pogacar stays humble with Monument history beckoning
Pogacar stays humble with Monument history beckoning
-
Real Madrid hoping Champions League magic halts Bayern juggernaut

-
 Sputtering Arsenal face test of character in Sporting clash
Sputtering Arsenal face test of character in Sporting clash
-
'Not the Cairo we know': Energy shock from Iran war dims Egypt nights

-
 Tokyo, Seoul shares gain, war sends oil higher
Tokyo, Seoul shares gain, war sends oil higher
-
Artemis mission headed for first lunar flyby since 1972

-
 South Korea president says regrets 'reckless' drones sent to North
South Korea president says regrets 'reckless' drones sent to North
-
Coughlin captures third LPGA title at Aramco Championship

-
 What to know about the Artemis 2 mission's Moon flyby
What to know about the Artemis 2 mission's Moon flyby
-
Mystique of the green jacket endures as Masters looms

-
 In El Salvador's mass trials, 'the innocent pay for the guilty'
In El Salvador's mass trials, 'the innocent pay for the guilty'
-
Trump makes stark threat to Iran after US airman rescued

-
 Trumps FDA CBD Enforcement Shift Signals a Turning Point - Why MMJ International Holdings is the Sector's Opportunity
Trumps FDA CBD Enforcement Shift Signals a Turning Point - Why MMJ International Holdings is the Sector's Opportunity
-
Amphastar Pharmaceuticals to Present at the 25th Annual Needham Healthcare Conference

-
 Texas Gulf Bank, N.A. Appoints Chase Zalman President
Texas Gulf Bank, N.A. Appoints Chase Zalman President
-
Bioz and Vilber Advance Evidence-Driven Scientific Marketing with Custom Publication Integration

-
 Artemis astronauts ready for Moon flyby on fifth day of historic mission
Artemis astronauts ready for Moon flyby on fifth day of historic mission
-
Israel renews Lebanon strikes, forces Syria border crossing closed

-
 Eagle-eyed Spaun snatches Texas Open victory
Eagle-eyed Spaun snatches Texas Open victory
-
Brown, Tatum propel Celtics in win over Raptors

-
 Paul battles past Burruchaga to win ATP Houston title
Paul battles past Burruchaga to win ATP Houston title
-
Major sponsors drop Kanye West London gigs as PM voices concern

-
 Inter close in on Serie A title by thumping Roma
Inter close in on Serie A title by thumping Roma
-
Trump makes foul-mouthed threat to Iran after US airman rescued

-
 Monaco sink Marseille for seventh Ligue 1 win in a row
Monaco sink Marseille for seventh Ligue 1 win in a row
-
Inter thump Roma to extend Serie A lead to nine points

-
 Lebanon's Christians mark Easter in solidarity with war-hit south
Lebanon's Christians mark Easter in solidarity with war-hit south
-
Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years

-
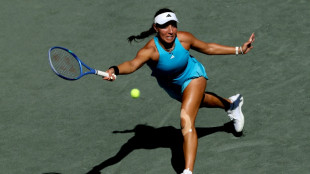 Pegula romps to WTA Charleston Open victory
Pegula romps to WTA Charleston Open victory
-
David six-hitting spree powers Bengaluru to IPL win

-
 Union draw leaves St Pauli stranded in Bundesliga drop zone
Union draw leaves St Pauli stranded in Bundesliga drop zone
-
UK police arrest protesters near base used by US

-
 Trump issues foul-mouthed threat to Iran after US airman rescued
Trump issues foul-mouthed threat to Iran after US airman rescued
-
Alcaraz plans to play full clay-court season, get 'socks dirty'

-
 'Super Mario Galaxy' blasts off in N. America box office debut
'Super Mario Galaxy' blasts off in N. America box office debut
-
Artemis astronauts begin fifth day on historic Moon mission

-
 Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup
Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup
-
Trump draws criticism with fiery Easter message on Iran

-
 OPEC+ hikes oil production quotas, issues warning
OPEC+ hikes oil production quotas, issues warning
-
British PM slams London event for booking Kanye West, sponsor quits


Ayrmid Reports Additional New Real-World Data on Motixafortide for Stem Cell Mobilization in Sickle Cell Disease and Beta-Thalassemia
Key Highlights
Motixafortide effectively mobilized sufficient hematopoietic stem cells (HSCs) in patients with sickle cell disease and beta-thalassemia enabling accelerated access to gene therapies
73% (11 of 15) of patients were able to collect sufficient HSC to enable progress to gene therapy manufacturing with 5 patients receiving their gene therapy and appropriately engrafted
73% (11 of 15) of patients had previously failed to collect sufficient cells with plerixafor
Results support the use of motixafortide as an effective single-agent mobilizer of HSC for people with sickle cell disease and in combination with G-CSF for beta-thalassemia
Findings presented at TANDEM 2026
DUBLIN, IE / ACCESS Newswire / February 4, 2026 / Ayrmid, Ltd. ("Ayrmid" or the "Company"), the parent company of Gamida Cell Inc., today announced further encouraging real-world data on the use of motixafortide, a long-acting CXCR4 inhibitor licensed by Ayrmid under the brand name APHEXDA®, for mobilizing HSCs in patients with sickle cell disease undergoing gene therapy. The results were presented at the 2025 Transplantation & Cellular Therapy meetings of ASTCT® and CIBMTR® (TANDEM), taking place from February 4-7, 2026 in Salt Lake City, UT.
Motixafortide is currently FDA approved in combination with filgrastim (G-CSF) for stem cell mobilization in multiple myeloma. The product's effectiveness and pharmacologic profile have prompted growing interest in its potential use for sickle cell disease, where collecting adequate stem cells remains a significant barrier to gene therapy. Many patients do not mobilize enough cells with standard approaches, limiting access to curative treatment.
Researchers from five treatment centers evaluated the real-world use of motixafortide in 15 patients aged 14-50. Each patient underwent one or more collection cycles over two or more days to obtain the number of stem cells required for gene therapy manufacturing. Motixafortide effectively mobilized 11 patients who had previously failed plerixafor, 3 patients mobilized effectively with motixafortide used as first line mobilization therapy.
In 11 patients, sufficient cells were achieved to manufacture gene therapy for both sickle cell disease and beta-thalassemia. 5 patients have received their gene therapy and have appropriately engrafted, manufacturing is underway in a further 5 patients. Patients received Casgevy and Lyfgenia, and manufacturing is underway for Casgevy, Lyfgenia and Zynteglo. This data supports that motixafortide mobilized cells can be used across different gene therapy manufacturing platforms.
Dr. John Manis,Director Transfusion Medicine Service, Associate Professor of Pathology, Harvard Medical School, commented: "The approval of gene therapies for sickle cell disease has opened the door to transformative outcomes, but inadequate stem cell collection has significantly delayed manufacturing and prevented some patients from starting treatment. These findings suggest that motixafortide may help overcome this challenge, and with more studies, expand access to potentially life-changing therapies."
About Sickle Cell Disease
Sickle cell disease, also known as sickle cell anemia, is a severe inherited blood disorder caused by a genetic mutation that leads to misshapen, rigid red blood cells. These cells can obstruct blood flow, causing sudden episodes of severe pain, known as pain crises, and leading to life-threatening complications. Ayrmid is dedicated to advancing therapies that help individuals living with sickle cell disease manage their symptoms and improve their quality of life.
About Beta-Thalassemia
Beta-thalassemia is a genetic blood disorder characterized by reduced or absent production of beta-globin, a component of hemoglobin, which is essential for carrying oxygen in red blood cells. This condition results in anemia, which can cause symptoms such as fatigue, weakness and pallor.
About Ayrmid Ltd. and Gamida Cell
Ayrmid Ltd. is the parent company of Gamida Cell Inc., a pioneering cell therapy company developing novel treatments designed to turn cells into powerful therapeutics. Gamida Cell Inc. currently has two FDA approved products on the market in the US, namely Omisirge® (please see the current full Prescribing Information, including boxed warning, here) and APHEXDA® (please see the current full Prescribing Information here). Gamida Cell operates as a wholly owned subsidiary of Ayrmid Limited, a UK entity. For additional information, please visit www.gamida-cell.com or follow Gamida Cell on LinkedIn, X, Facebook or Instagram.
Contacts: Media, Investors / Business Development: [email protected]
This information is provided by RNS, the news service of the London Stock Exchange. RNS is approved by the Financial Conduct Authority to act as a Primary Information Provider in the United Kingdom. Terms and conditions relating to the use and distribution of this information may apply. For further information, please contact [email protected] or visit www.rns.com.
SOURCE: Ayrmid Pharma Ltd
View the original press release on ACCESS Newswire
R.Lee--AT
