
-
 Swimmers McKeown, O'Callaghan and Chalmers dominate at Australian Open
Swimmers McKeown, O'Callaghan and Chalmers dominate at Australian Open
-
Bucha: When the Russian killers came...

-
 Iran, a Terrorist State with No Right to Exist
Iran, a Terrorist State with No Right to Exist
-
African players in Europe: Semenyo scores as City rout Liverpool

-
 Israeli strikes kill Iran Guards intel chief as Trump deadline looms
Israeli strikes kill Iran Guards intel chief as Trump deadline looms
-
Saving energy in everyday life or a complete rip-off?

-
 US sprint star Richardson wins Australia's Stawell Gift in record time
US sprint star Richardson wins Australia's Stawell Gift in record time
-
Rockets down Warriors in Curry return, Flagg carries Mavs past Lakers

-
 Artemis mission approaches lunar loop for first flyby since 1972
Artemis mission approaches lunar loop for first flyby since 1972
-
Israeli rescuers search for missing in building strike, two dead

-
 Defiant Iran ramps up attacks after Trump warning
Defiant Iran ramps up attacks after Trump warning
-
Saudi oasis town adjusts to life in the firing line

-
 Pogacar stays humble with Monument history beckoning
Pogacar stays humble with Monument history beckoning
-
Real Madrid hoping Champions League magic halts Bayern juggernaut

-
 Sputtering Arsenal face test of character in Sporting clash
Sputtering Arsenal face test of character in Sporting clash
-
'Not the Cairo we know': Energy shock from Iran war dims Egypt nights

-
 Tokyo, Seoul shares gain, war sends oil higher
Tokyo, Seoul shares gain, war sends oil higher
-
Artemis mission headed for first lunar flyby since 1972

-
 South Korea president says regrets 'reckless' drones sent to North
South Korea president says regrets 'reckless' drones sent to North
-
Coughlin captures third LPGA title at Aramco Championship

-
 What to know about the Artemis 2 mission's Moon flyby
What to know about the Artemis 2 mission's Moon flyby
-
Mystique of the green jacket endures as Masters looms

-
 In El Salvador's mass trials, 'the innocent pay for the guilty'
In El Salvador's mass trials, 'the innocent pay for the guilty'
-
Trump makes stark threat to Iran after US airman rescued

-
 Bora Biologics Successfully Completes 2,000L Engineering and Scale-Up Run in San Diego, Reinforcing Commercial-Scale Readiness
Bora Biologics Successfully Completes 2,000L Engineering and Scale-Up Run in San Diego, Reinforcing Commercial-Scale Readiness
-
Avino Announces Normal Course Issuer Bid for Common Shares

-
 Revolve Signs Interconnection Agreement for 130 MW EL 24 Wind Project in Mexico
Revolve Signs Interconnection Agreement for 130 MW EL 24 Wind Project in Mexico
-
Building the Moon's Future: Helio Positioned at the Center of America's Lunar Strategy for the Dawn of the Artemis Era

-
 Trumps FDA CBD Enforcement Shift Signals a Turning Point - Why MMJ International Holdings is the Sector's Opportunity
Trumps FDA CBD Enforcement Shift Signals a Turning Point - Why MMJ International Holdings is the Sector's Opportunity
-
Amphastar Pharmaceuticals to Present at the 25th Annual Needham Healthcare Conference

-
 Texas Gulf Bank, N.A. Appoints Chase Zalman President
Texas Gulf Bank, N.A. Appoints Chase Zalman President
-
Bioz and Vilber Advance Evidence-Driven Scientific Marketing with Custom Publication Integration

-
 Artemis astronauts ready for Moon flyby on fifth day of historic mission
Artemis astronauts ready for Moon flyby on fifth day of historic mission
-
Israel renews Lebanon strikes, forces Syria border crossing closed

-
 Eagle-eyed Spaun snatches Texas Open victory
Eagle-eyed Spaun snatches Texas Open victory
-
Brown, Tatum propel Celtics in win over Raptors

-
 Paul battles past Burruchaga to win ATP Houston title
Paul battles past Burruchaga to win ATP Houston title
-
Major sponsors drop Kanye West London gigs as PM voices concern

-
 Inter close in on Serie A title by thumping Roma
Inter close in on Serie A title by thumping Roma
-
Trump makes foul-mouthed threat to Iran after US airman rescued

-
 Monaco sink Marseille for seventh Ligue 1 win in a row
Monaco sink Marseille for seventh Ligue 1 win in a row
-
Inter thump Roma to extend Serie A lead to nine points

-
 Lebanon's Christians mark Easter in solidarity with war-hit south
Lebanon's Christians mark Easter in solidarity with war-hit south
-
Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years

-
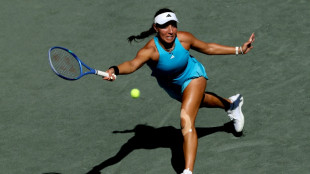 Pegula romps to WTA Charleston Open victory
Pegula romps to WTA Charleston Open victory
-
David six-hitting spree powers Bengaluru to IPL win

-
 Union draw leaves St Pauli stranded in Bundesliga drop zone
Union draw leaves St Pauli stranded in Bundesliga drop zone
-
UK police arrest protesters near base used by US

-
 Trump issues foul-mouthed threat to Iran after US airman rescued
Trump issues foul-mouthed threat to Iran after US airman rescued
-
Alcaraz plans to play full clay-court season, get 'socks dirty'


IGC Pharma Reaches 70% Enrollment in Phase 2 CALMA Trial Evaluating IGC-AD1 for Alzheimer's Agitation
POTOMAC, MARYLAND / ACCESS Newswire / February 2, 2026 / IGC Pharma, Inc. (NYSE American:IGC) ("IGC" or the "Company"), today reported it has reached approximately 70% of planned patient enrollment in its Phase 2 CALMA clinical trial evaluating IGC-AD1 for the treatment of agitation associated with Alzheimer's disease.
The Company has approximately 23 active sites across 26 locations, and enrollment is progressing across this clinical network. IGC expects to complete enrollment by mid-2026. The Company believes this milestone meaningfully reduces the primary operational risk for the trial and positions CALMA to progress toward database lock and topline results.
"Reaching ~70% enrollment reflects improving execution and site productivity across our network," said Ram Mukunda, CEO of IGC Pharma. "Enrollment is the final operational gate, and our focus is straightforward: complete enrollment efficiently while maintaining data quality and rigorous trial conduct."
Operational Updates
The Company has continued to activate experienced sites and expand patient access through a hybrid, decentralized model in select geographies. IGC expects enrollment progress to continue as additional site capacity comes online, and newly activated sites ramp to steady-state contribution. In a randomized, double-blind, placebo-controlled study, efficacy outcomes are not analyzed on an ongoing interim basis to preserve blinding and trial integrity.
IGC-AD1 is an investigational, cannabinoid-based therapy being evaluated in a randomized, double-blind, placebo-controlled clinical trial designed to assess safety and efficacy in agitation associated with Alzheimer's disease.
About IGC Pharma (dba IGC):
IGC Pharma (NYSE American: IGC) is a clinical-stage biotechnology company leveraging AI to develop innovative treatments for Alzheimer's and metabolic disorders. Our lead asset, IGC-AD1, is a cannabinoid-based therapy currently in a Phase 2 trial (CALMA) for agitation in Alzheimer's dementia. Our pipeline includes TGR-63, targeting amyloid plaques, and early-stage programs focused on neurodegeneration, tau proteins, and metabolic dysfunctions. We integrate AI to accelerate drug discovery, optimize clinical trials, and enhance patient targeting. With a complete patent portfolio and a commitment to innovation, IGC Pharma is advancing breakthrough therapies.
Forward-Looking Statements:
This press release contains forward-looking statements. These forward-looking statements are based largely on IGC Pharma's expectations and are subject to several risks and uncertainties, certain of which are beyond IGC Pharma's control. Actual results could differ materially from these forward-looking statements as a result of, among other factors, the Company's failure or inability to commercialize one or more of the Company's products or technologies, including the products or formulations described in this release, or failure to obtain regulatory approval for the products or formulations, where required, or government regulations affecting AI or the AI algorithms not working as intended or producing accurate predictions; general economic conditions that are less favorable than expected; the FDA's general position regarding cannabis- and hemp-based products; and other factors, many of which are discussed in IGC Pharma's U.S. Securities and Exchange Commission ("SEC") filings. IGC incorporates by reference its Annual Report on Form 10-K filed with the SEC on June 27, 2025, as if fully incorporated and restated herein. Considering these risks and uncertainties, there can be no assurance that the forward-looking information contained in this release will occur. IGC Pharma, Inc. assumes no obligation to update forward-looking statements contained in this release as the result of new information or future events or developments.
Contact Information:
Rosalyn Christian / John Nesbett
IMS Investor Relations
[email protected]
(203) 972-9200
SOURCE: IGC Pharma, Inc.
View the original press release on ACCESS Newswire
W.Moreno--AT
