
-
 Injured Bayern starlet Karl to miss Real return leg
Injured Bayern starlet Karl to miss Real return leg
-
US-Iran talks in Pakistan uncertain as sides trade accusations

-
 Oil spill snarls shipping traffic in Antwerp port
Oil spill snarls shipping traffic in Antwerp port
-
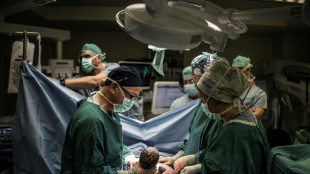
Giving birth in a shelter in Israel
-
 Five things to know about the planned Iran-US talks in Islamabad
Five things to know about the planned Iran-US talks in Islamabad
-
Slot feels 'complete support' from Liverpool chiefs despite slump

-
 Kyiv books tentative diplomatic coup with Iran war forays
Kyiv books tentative diplomatic coup with Iran war forays
-
Teenager shines as Britain seize control of BJK Cup tie with Australia

-
 Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader
Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader
-
Sleepy seal diverts traffic in Australian seaside town

-
 Artemis astronauts to shed light on space health risks
Artemis astronauts to shed light on space health risks
-
Pakistan prepares to host US-Iran talks, as Lebanon fighting continues

-
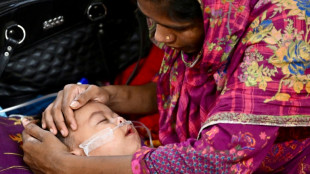 Vaccine gaps fuel Bangladesh's deadly measles crisis
Vaccine gaps fuel Bangladesh's deadly measles crisis
-
Fish furore fuels fierce election in India's West Bengal

-
 Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G
Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G
-
Myanmar junta chief sworn in as president

-
 Exiled cartoonists give voice to Iran's silenced millions
Exiled cartoonists give voice to Iran's silenced millions
-
In Pakistan's mediation to end Mideast war, China may hold the key

-
 Knicks stay in hunt with late win over rival Celtics
Knicks stay in hunt with late win over rival Celtics
-
'Sartorial diplomacy' on show in expo of late UK queen's fashion

-
 Former Japan and AC Milan star Honda laces up boots again at 39
Former Japan and AC Milan star Honda laces up boots again at 39
-
Stocks rally on optimism over Iran war ceasefire, oil extends gains

-
 Lego-style memes troll Trump after fragile US-Iran truce
Lego-style memes troll Trump after fragile US-Iran truce
-
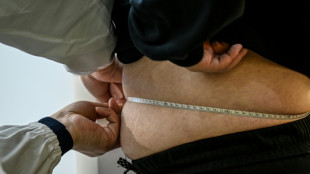
Chinese slimmers trade lost fat for beef
-
 Jackson biopic shows franchise thriving despite abuse claims
Jackson biopic shows franchise thriving despite abuse claims
-
New Jersey city spurns data center as defiance spreads

-
 US box office looking good as cinema owners gather: industry chief
US box office looking good as cinema owners gather: industry chief
-
Firm Masters greens make life hard on golf's finest

-
 GMV Minerals Announces Drill Mobilization at the Mexican Hat Gold Deposit in S.E. Arizona
GMV Minerals Announces Drill Mobilization at the Mexican Hat Gold Deposit in S.E. Arizona
-
Gamma Resources Adopts Semi-Annual Financial Reporting

-
 Defending champ McIlroy shares Masters lead after back-nine birdie run
Defending champ McIlroy shares Masters lead after back-nine birdie run
-
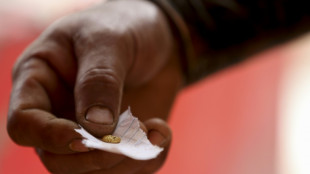
After oil, Venezuela opens up mining to private investors
-
 Tigers' Meadows in hospital after colliding with teammate
Tigers' Meadows in hospital after colliding with teammate
-
US to host Israel-Lebanon talks as strikes threaten Iran ceasefire

-
 'Scrappy' McIlroy leans on experience for share of Masters lead
'Scrappy' McIlroy leans on experience for share of Masters lead
-
Ukraine and Russia will cease fire for Orthodox Easter

-
 Mateta inspires Palace win over Fiorentina in Conference League
Mateta inspires Palace win over Fiorentina in Conference League
-
Pioneering US hip-hop artist Afrika Bambaataa dies at 68

-
 Russia bans Nobel-winning rights group, raids independent newspaper, in one day
Russia bans Nobel-winning rights group, raids independent newspaper, in one day
-
Pentagon denies giving Vatican envoy 'bitter lecture'

-
 Watkins propels Villa towards Europa League semis, Forest hold Porto
Watkins propels Villa towards Europa League semis, Forest hold Porto
-
Aston Villa on verge of Europa League semis after beating Bologna

-
 Venezuela police clash with protesters demanding salary rises
Venezuela police clash with protesters demanding salary rises
-
CAF president rejects corruption claims by Senegal

-
 Israel and Lebanon set for ceasefire talks next week, says US official
Israel and Lebanon set for ceasefire talks next week, says US official
-
US stocks extend gains, shrugging off ceasefire worries

-
 IMF chief urges nations to 'do no harm' in fiscal response to Iran war
IMF chief urges nations to 'do no harm' in fiscal response to Iran war
-
Sixers' Embiid to have surgery for appendicitis - team

-
 Russian police raid independent Novaya Gazeta outlet, reporter detained
Russian police raid independent Novaya Gazeta outlet, reporter detained
-
Former heavyweight king Fury adamant 'I've still got it' as Makhmudov awaits


One-Year Data from the VenoValve(R) U.S. Pivotal Trial Highlighting Impact on Patients' Quality of Life (QOL) to be Presented Today at the 37th Annual Meeting of the American Venous Forum
One-year follow-up data demonstrates sustained improvements across venous specific quality-of-life (QoL) indicators.
PMA application for the VenoValve submitted in November 2024, with an FDA decision expected in the second half of 2025.
IRVINE, CA / ACCESS Newswire / February 19, 2025 / enVVeno Medical Corporation (NASDAQ:NVNO) ("enVVeno" or the "Company"), a company setting new standards of care for the treatment of venous disease, today announced that one-year follow-up data on 75 subjects from the VenoValve U.S. pivotal trial will be presented by Dr. Cassius Iyad Ochoa Chaar, Associate Professor Surgery, Yale School of Medicine, Division of Vascular and Endovascular Surgery, Principal Investigator, and lead enroller for the trial, at the 37th Annual Meeting of the American Venous Forum (VENOUS2025) being held February 16-19, 2025 in Atlanta, GA. The presentation is scheduled for today at 9:47 AM ET.

The data show that trial subjects who had the SAVVE procedure experienced statistically significant improvements in QoL metrics related to venous disease at 12 months compared to baseline as demonstrated by the VEINS-SYM/QOL scores. To view results from the trial, please visit www.venovalve.com. The Company has submitted a pre-market authorization (PMA) application for the VenoValve to the U.S. Food and Drug Administration (FDA), with a decision anticipated in the second half of 2025.
"We continue to be asked to present our data at leading vascular conferences throughout the world as news of the potential for the VenoValve spreads among vascular surgeons whose practices include the treatment of venous diseases," said Robert Berman, enVVeno Medical's Chief Executive Officer. "The VenoValve has the potential to be the first FDA approved treatment for deep venous CVI, and we are attempting to reach the top of a mountain that nobody has successfully climbed and with many failed attempts over the past several decades. With the summit of the mountain finally in sight, this is an exciting time for our company and for the millions of patients with severe deep venous CVI who have no effective treatment options."
Severe, deep venous CVI is a debilitating disease that is most often caused by blood clots (deep vein thromboses or DVTs) in the deep veins of the leg. When valves inside of the veins of the leg fail, blood flows in the wrong direction and pools in the lower leg, causing pressure within the veins of the leg to increase (venous hypertension). Symptoms of severe CVI include leg swelling, pain, edema, and in the most severe cases, recurrent open sores known as venous ulcers. The disease can severely impact everyday functions such as sleeping, bathing, dressing, and walking, and is known to result in high rates of depression and anxiety. There are currently no effective treatments for severe CVI of the deep vein system caused by valvular incompetence. Estimates indicate that CVI costs the U.S. healthcare system in excess of $4 billion each year.
The Venous Insufficiency Epidemiological and Economic Study Quality of Life/Symptoms (VEINES-QoL/Sym) questionnaire is a validated, disease-specific instrument to measure patient-reported quality of life and symptom severity in individuals with chronic venous disease. By assessing factors such as pain, functional limitations, and psychosocial impacts, VEINES-QoL/Sym provides a standardized framework for evaluating treatment outcomes and guiding clinical decision-making.
About enVVeno Medical Corporation
enVVeno Medical (NASDAQ:NVNO) is an Irvine, California-based, late clinical-stage medical device Company focused on the advancement of innovative bioprosthetic (tissue-based) solutions to improve the standard of care for the treatment of venous disease. The Company's lead product, the VenoValve®, is a first-in-class surgical replacement venous valve being developed for the treatment of deep venous Chronic Venous Insufficiency (CVI). The Company is also developing a non-surgical, transcatheter based replacement venous valve for the treatment of deep venous CVI called enVVe®. CVI occurs when valves inside of the veins of the leg become damaged, resulting in the backwards flow of blood (reflux), blood pooling in the lower leg, increased pressure in the veins of the leg (venous hypertension) and in severe cases, venous ulcers that are difficult to heal and become chronic. Both the VenoValve and enVVe are designed to act as one-way valves, to help assist in propelling blood up the leg, and back to the heart and lungs. The VenoValve is currently being evaluated in the SAVVE U.S. pivotal study and the Company is currently performing the final testing necessary to seek approval for the pivotal trial for enVVe.
Cautionary Note on Forward-Looking Statements
This press release and any statements of stockholders, directors, employees, representatives and partners of enVVeno Medical Corporation (the "Company") related thereto contain, or may contain, among other things, certain "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements involve significant risks and uncertainties. Such statements may include, without limitation, statements identified by words such as "projects," "may," "will," "could," "would," "should," "believes," "expects," "anticipates," "estimates," "intends," "plans," "potential" or similar expressions. These statements are based upon the current beliefs and expectations of the Company's management and are subject to significant risks and uncertainties, including those detailed in the Company's filings with the Securities and Exchange Commission. Actual results and timing (may differ significantly from those set forth or implied in the forward-looking statements. Forward-looking statements involve certain risks and uncertainties that are subject to change based on various factors (many of which are beyond the Company's control). The Company undertakes no obligation to publicly update any forward-looking statements, whether as a result of new information, future presentations or otherwise, except as required by applicable law.
###
INVESTOR CONTACT:
Jenene Thomas, JTC Team, LLC
[email protected]
(908) 824-0775
SOURCE: enVVeno Medical Corporation
View the original press release on ACCESS Newswire
Ch.P.Lewis--AT


