
-
 Exiled cartoonists give voice to Iran's silenced millions
Exiled cartoonists give voice to Iran's silenced millions
-
In Pakistan's mediation to end Mideast war, China may hold the key

-
 Knicks stay in hunt with late win over rival Celtics
Knicks stay in hunt with late win over rival Celtics
-
'Sartorial diplomacy' on show in expo of late UK queen's fashion

-
 Former Japan and AC Milan star Honda laces up boots again at 39
Former Japan and AC Milan star Honda laces up boots again at 39
-
Stocks rally on optimism over Iran war ceasefire, oil extends gains

-
 Lego-style memes troll Trump after fragile US-Iran truce
Lego-style memes troll Trump after fragile US-Iran truce
-
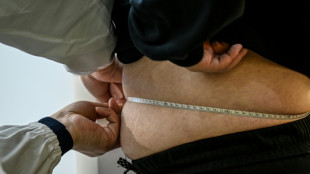
Chinese slimmers trade lost fat for beef
-
 Jackson biopic shows franchise thriving despite abuse claims
Jackson biopic shows franchise thriving despite abuse claims
-
New Jersey city spurns data center as defiance spreads

-
 US box office looking good as cinema owners gather: industry chief
US box office looking good as cinema owners gather: industry chief
-
Firm Masters greens make life hard on golf's finest

-
 Defending champ McIlroy shares Masters lead after back-nine birdie run
Defending champ McIlroy shares Masters lead after back-nine birdie run
-
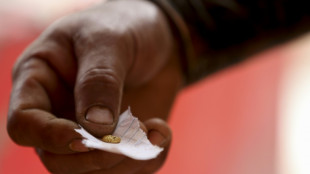
After oil, Venezuela opens up mining to private investors
-
 Tigers' Meadows in hospital after colliding with teammate
Tigers' Meadows in hospital after colliding with teammate
-
US to host Israel-Lebanon talks as strikes threaten Iran ceasefire

-
 'Scrappy' McIlroy leans on experience for share of Masters lead
'Scrappy' McIlroy leans on experience for share of Masters lead
-
Ukraine and Russia will cease fire for Orthodox Easter

-
 Mateta inspires Palace win over Fiorentina in Conference League
Mateta inspires Palace win over Fiorentina in Conference League
-
Pioneering US hip-hop artist Afrika Bambaataa dies at 68

-
 Russia bans Nobel-winning rights group, raids independent newspaper, in one day
Russia bans Nobel-winning rights group, raids independent newspaper, in one day
-
Pentagon denies giving Vatican envoy 'bitter lecture'

-
 Watkins propels Villa towards Europa League semis, Forest hold Porto
Watkins propels Villa towards Europa League semis, Forest hold Porto
-
Aston Villa on verge of Europa League semis after beating Bologna

-
 Venezuela police clash with protesters demanding salary rises
Venezuela police clash with protesters demanding salary rises
-
CAF president rejects corruption claims by Senegal

-
 Israel and Lebanon set for ceasefire talks next week, says US official
Israel and Lebanon set for ceasefire talks next week, says US official
-
US stocks extend gains, shrugging off ceasefire worries

-
 IMF chief urges nations to 'do no harm' in fiscal response to Iran war
IMF chief urges nations to 'do no harm' in fiscal response to Iran war
-
Sixers' Embiid to have surgery for appendicitis - team

-
 Russian police raid independent Novaya Gazeta outlet, reporter detained
Russian police raid independent Novaya Gazeta outlet, reporter detained
-
Former heavyweight king Fury adamant 'I've still got it' as Makhmudov awaits

-
 Shipping toll for Hormuz passage sharply divides nations
Shipping toll for Hormuz passage sharply divides nations
-
McIlroy's back-nine birdie run grabs share of Masters lead

-
 Melania Trump blasts 'lies' linking her to Epstein
Melania Trump blasts 'lies' linking her to Epstein
-
'Anxious' Tatum back at Madison Square Garden with NBA East second seed on line

-
 Strait of Hormuz traffic remains becalmed despite ceasefire
Strait of Hormuz traffic remains becalmed despite ceasefire
-
Melania Trump denies any links to Epstein abuse

-
 American Airlines targets April 30 return to Venezuela
American Airlines targets April 30 return to Venezuela
-
Venezuela police tear-gas protesters demanding salary rises

-
 Robertson to leave Liverpool at end of season
Robertson to leave Liverpool at end of season
-
Choudhary smashes Lucknow to dramatic IPL win over Kolkata

-
 Sean 'Diddy' Combs asks US appeals court to overturn sentence
Sean 'Diddy' Combs asks US appeals court to overturn sentence
-
Verstappen Red Bull future in doubt as engineer to join McLaren

-
 France's Macron in Rome for first meeting with Pope Leo
France's Macron in Rome for first meeting with Pope Leo
-
Angola name former Senegal boss Cisse as new coach

-
 Sinner and Alcaraz wobble but advance to Monte Carlo quarter-finals
Sinner and Alcaraz wobble but advance to Monte Carlo quarter-finals
-
Reed soars to early Masters lead on wings of eagles

-
 US Democrats fail in bid to curb Trump's Iran war powers
US Democrats fail in bid to curb Trump's Iran war powers
-
Veteran prop Slimani to return to France with Toulon


Adolore BioTherapeutics to Present CA8* Neuromodulating Gene Therapy at the 2025 NANS Annual Meeting
DELRAY BEACH, FL / ACCESS Newswire / February 4, 2025 / Adolore BioTherapeutics ("Adolore" or the "Company"), is developing intracellularly-delivered neuromodulators that naturally reduce neuronal excitability to restore balance to the PNS/CNS to treat the underlying root cause of serious neurological disorders, including chronic pain, today announced that Roy C. Levitt, MD, the Company's founder, Chief Medical Officer, and Executive Chairman, Clinical Professor, University of Miami, and Principal Investigator of a NIH, NINDS, HEAL UH3 Award will present at the 2025 North American Neuromodulation Society, ("NANS"), annual meeting being held January 30 - February 1, 2025 in Orlando, Fl.
"Neuromodulation using gene therapy is an exciting new area that has wide-spread applications in treating not only chronic pain but other serious undertreated neurological disorders. We are delighted to present our cutting-edge technology at NANS," commented Dr. Levitt. There is a paucity of safe, efficacious non-opioid analgesic treatments for chronic pain, creating a large and very urgent unmet medical need given the ongoing opioid crisis. Dr. Levitt will present Adolore's innovative CA8* chronic pain therapeutic (*carbonic anhydrase-like analgesic peptides, CA8 variants) and its cutting-edge technology using disease-free, nontoxic replication-defective Herpes Simplex Virus, ("rdHSV"), vectors. The company currently has two therapeutic programs in development, including peripheral neuropathy caused by erythromelalgia, which is an orphan disease, (ADB-101), and Adolore's lead program (ADB-102) treating patients with chronic pain caused by moderate-to-severe knee osteoarthritis. Based on compelling preclinical data, the Company is progressing these programs toward Investigational New Drug, ("IND"), filings and first-in-human clinical studies. These CA8* pain therapies were licensed in 2023 by Adolore from the University of Miami, Miami, FL. The rdHSV intracellular biotherapeutics delivery technology was licensed by Adolore from the University of Pittsburgh in 2023.
The Company's development program for the treatment of chronic pain due to moderate-to-severe knee osteoarthritis is supported by the NIH/NINDS HEAL UH3 Award to the University of Miami that funds all formal pre-clinical GLP/GMP/GCP development work through a first-in-human clinical study in patients, which is expected to commence in 2026.
For more information about the 2025 NANS Annual Meeting, visit the meeting website at: https://www.neuromodulation.org/annual-meeting-overview.html
About Adolore BioTherapeutics, Inc.
Adolore BioTherapeutics, Inc., is focused on developing novel intracellularly delivered treatments for chronic pain, drug-resistant epilepsy, seizure disorders, neurodegeneration and neurotrauma/neuroprotection, e.g., hearing loss. The Company's chronic pain treatment platform comprises two highly innovative technologies: our CA8* analgesic peptides, and our cutting-edge, disease-free, nontoxic rdHSV vector-based intracellular biotherapeutics delivery technology. Our best-in-class programs are long-acting, localized gene-therapies that are opioid-free Disease Modifying Anti-Pain therapies (DMAPs).
The Company's two current CA8* DMAP programs are in preclinical development for treating patients suffering from erythromelalgia, a life-long heritable chronic pain condition representing an orphan drug disease with no approved therapy, and patients suffering with chronic pain due to moderate-to-severe knee osteoarthritis, which affects a large number of patients that are often treated with opioids due to the lack of good alternatives, thus contributing to the ongoing opioid crisis.
For more information, visit adolore.com.
Forward Looking Statements
To the extent this announcement contains information and statements that are not historical, they are considered forward-looking statements within the meaning of the federal securities laws. You can identify forward-looking statements by the use of the words "believe," "expect," "anticipate," "intend," "estimate," "project," "will," "should," "may," "plan," "intend," "assume" and other expressions which predict or indicate future events and trends and which do not relate to historical matters. You should not rely on forward-looking statements, because they involve known and unknown risks, uncertainties and other factors, some of which are beyond the control of the Company. These risks and uncertainties include, but are not limited to, those associated with drug development. These risks, uncertainties and other factors may cause the actual results, performance or achievements of the Company to be materially different from the anticipated future results, performance or achievements expressed or implied by the forward-looking statements.
Investor Relations Contact
Paul Barone
(215)622-4542
[email protected]
SOURCE: Adolore Biotherapeutics, Inc.
View the original press release on ACCESS Newswire
W.Nelson--AT



