-
 Australian regulator probes Facebook, YouTube over teen social media ban
Australian regulator probes Facebook, YouTube over teen social media ban
-
Iraq coach shielding players from war ahead of World Cup bid

-
 Undav rescues Germany late in Ghana friendly
Undav rescues Germany late in Ghana friendly
-
Messi to start for Argentina in World Cup send-off: Scaloni

-
 Oil rises on Trump's Iran threats, stocks mixed
Oil rises on Trump's Iran threats, stocks mixed
-
After pope's remark, White House defends praying for US troops

-
 Powell probe leaves US Fed leadership change in limbo
Powell probe leaves US Fed leadership change in limbo
-
Celine Dion announces comeback following health struggle

-
 'Is it Kafka?' US judge baffled by new Pentagon press policy
'Is it Kafka?' US judge baffled by new Pentagon press policy
-
Cubans ready for Russian oil but some say not enough

-
 Teen Suryavanshi shines as Rajasthan hammer Chennai in IPL
Teen Suryavanshi shines as Rajasthan hammer Chennai in IPL
-
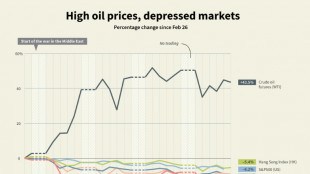
Stock market winners and losers one month into US-Israel war on Iran
-
 Hodgson says surprise return to management is only for short-term
Hodgson says surprise return to management is only for short-term
-
What could Trump achieve by threatening Iran's Kharg Island?

-
 India declares victory over Maoist insurgency
India declares victory over Maoist insurgency
-
Germany's Merz pushes return of Syrians as he hosts leader Sharaa

-
 G7 ministers pledge 'necessary measures' to ensure stable energy market
G7 ministers pledge 'necessary measures' to ensure stable energy market
-
Cardiff City lose compensation case over Emiliano Sala death

-
 Several French far-right mayors take down EU flags
Several French far-right mayors take down EU flags
-
Air Canada CEO to retire after row over English-only condolence message

-
 Oil rises on Trump's Iran threats, stocks take cue on talks
Oil rises on Trump's Iran threats, stocks take cue on talks
-
Syrian leader pledges to work with Germany on migration, recovery

-
 AI agent future is coming, OpenClaw creator tells AFP
AI agent future is coming, OpenClaw creator tells AFP
-
Cardiff lose 122 mn euro compensation case over Emiliano Sala death

-
 Tuchel defends Rice and Saka after England withdrawals
Tuchel defends Rice and Saka after England withdrawals
-
G7 ministers tackle economic fallout of Mideast war

-
 Tottenham close in on De Zerbi as next boss - reports
Tottenham close in on De Zerbi as next boss - reports
-
Kenya's former NY marathon champion Korir gets 5-year doping ban

-
 Lukaku says 'could never turn back on Napoli' after treatment row
Lukaku says 'could never turn back on Napoli' after treatment row
-
Syrian leader visits Germany to talk war, recovery, refugees

-
 Renault says developing ground-based military drone
Renault says developing ground-based military drone
-
Iran hangs two 'political prisoners' from banned opposition: activists

-
 Russia expels UK diplomat on spying allegations
Russia expels UK diplomat on spying allegations
-
Premier League fans back call to scrap VAR

-
 Italy hoping to scale World Cup 'Everest' ahead of Bosnia play-off showdown
Italy hoping to scale World Cup 'Everest' ahead of Bosnia play-off showdown
-
Japan's cherry blossom season dazzles locals and tourists

-
 EU ups mackerel quotas to match UK despite overfishing concerns
EU ups mackerel quotas to match UK despite overfishing concerns
-
Crude rises, stocks drop as Houthi attacks escalate Iran war

-
 Australian Rules player banned for wiping blood on face of opponent
Australian Rules player banned for wiping blood on face of opponent
-
Sheep culls put pressure on Greek feta cheese production

-
 One man, his dog, and ChatGPT: Australia's AI vaccine saga
One man, his dog, and ChatGPT: Australia's AI vaccine saga
-
Israel PM restores access after Latin Patriarch blocked from Holy Sepulchre

-
 Israel strikes Tehran as Trump says Iran deal may be reached 'soon'
Israel strikes Tehran as Trump says Iran deal may be reached 'soon'
-
Italy chase World Cup spot as Kosovo bid to make debut

-
 Myanmar paves way for junta chief to become civilian president
Myanmar paves way for junta chief to become civilian president
-
'Long live the shah': Iranian diaspora back war at Washington rally

-
 Taiwan opposition leader accepts Xi's invitation to visit China
Taiwan opposition leader accepts Xi's invitation to visit China
-
French masonic lodge at heart of murky murder trial

-
 US military building 'massive complex' beneath White House ballroom project: Trump
US military building 'massive complex' beneath White House ballroom project: Trump
-
IPL captain takes pop at Cricket Australia over record-buy Green


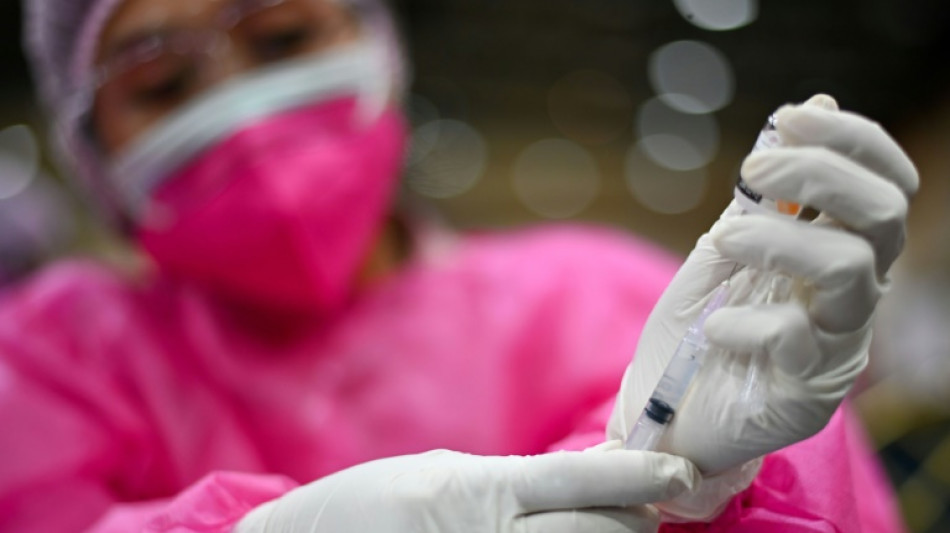
Indonesia approves first homegrown Covid-19 vaccine
Indonesia has approved its first locally developed Covid-19 vaccine for emergency use, the head of the country's public health agency said Friday, hailing it as a step toward "the nation's independence in access to medicine".
Jakarta has stressed the importance of developing national vaccines since the beginning of the pandemicbut it currently relies on China's Sinovac and the Western-made Moderna and Pfizer-BioNTech mRNA jabs.
The IndoVac jab, developed by state-owned pharmaceutical company Bio Farma and Texas-based Baylor College of Medicine, can now be used as a primary dose for an unvaccinated or partially vaccinated adult in the world's fourth most-populous country.
"The development... of a domestic vaccine is a pride for us Indonesians as a foundation and as the first step to achieve the nation's independence in access to medicine," head of the national food and drugs agency (BPOM) Penny Lukito said at a press conference Friday.
The medical chief said IndoVac had shown an efficacy rate of 92 percent, while there were no reports of death linked to it in trials and reported side effects were "generally mild".
A clinical study to use it as a booster jab is underway, Lukito said.
The agency also announced it had granted emergency use approval for an mRNA vaccine developed in China, becoming the first country to do so.
The inoculation developed by Walvax Biotechnology will be locally produced in Indonesia, the BPOM chief said.
The homegrown IndoVac jab has been granted a halal certificate, meaning it can be administered in line with the Islamic faith in Muslim-majority Indonesia.
Indonesia became the epicentre of Asia's Covid-19 pandemic in July last year as the Delta variant swept through the country.
Daily cases declined significantly by the end of the year but the spread of Omicron brought confirmed cases back to 30,000 a day.
It has since seen another sharp fall in case numbers andeliminated quarantine requirements for vaccinated travellers.
In total,Southeast Asia's largest economy has reported over 6.4 million confirmed cases with nearly 160,000 deaths.
Vaccination rollout has also been relatively slow compared to developed nations, with less than two-thirds of the population of 270 million receiving two jabs.
A.Moore--AT



