-
 Denmark, Australia in the spotlight in Eurovision second semi
Denmark, Australia in the spotlight in Eurovision second semi
-
Heavy Russian strikes on Kyiv kill one, wound 31

-
 Xi warns Trump on Taiwan at Beijing summit
Xi warns Trump on Taiwan at Beijing summit
-
Iran war and oil dominate BRICS meet in India

-
 Bone appetit: Paris pups lap up treats at dog-centric spots
Bone appetit: Paris pups lap up treats at dog-centric spots
-
Kohli senses end after roaring back to form with IPL century

-
 India bars sugar exports until September
India bars sugar exports until September
-
Madonna, Shakira, BTS to headline first World Cup final half-time show

-
 Japan takes 'half step' toward fixing slow retrial system
Japan takes 'half step' toward fixing slow retrial system
-
Honda posts operating loss, first since 1957

-
 Madonna, Shakira, BTS to headline World Cup final half-time show
Madonna, Shakira, BTS to headline World Cup final half-time show
-
A quarter of World Cup games risk searing heat: scientists

-
 Six hantavirus cruise passengers head to Australia
Six hantavirus cruise passengers head to Australia
-
Suspect detained in Philippine senate gunfire: police

-
 Cavs top Pistons in overtime for 3-2 series lead
Cavs top Pistons in overtime for 3-2 series lead
-
Canadian football ready for World Cup coming out party

-
 US court suspends sanctions on UN expert on Palestinians
US court suspends sanctions on UN expert on Palestinians
-
Asia markets mixed as Trump-Xi summit, AI trade dominate

-
 'Promised to us': The Israelis dreaming of settling south Lebanon
'Promised to us': The Israelis dreaming of settling south Lebanon
-
'Rare, meaningful': North Korean football team ventures into South

-
 In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
-
Historic Swiss solar-powered plane crashes into sea

-
 A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
-
Indian pharma fuels Africa's 'zombie drug' and opioid crisis

-
 After months of blackout, Iran gives internet to select few
After months of blackout, Iran gives internet to select few
-
Wood urges New Zealand to 'create some history' at World Cup

-
 In Washington, the fight to preserve Black cemeteries
In Washington, the fight to preserve Black cemeteries
-
US children's book author sentenced to life after poisoning husband

-
 Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
-
Battery X Metals Achieves Milestone with Delivery of Next-Generation Patent-Pending Lithium-Ion Battery Rebalancing Machine Featuring Design Enhancements, Advancing Strategic Commercialization Initiatives

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14
InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14
-
Akkodis Recognized in HFS Horizons 2026 Report for Enterprise Ready Agentic AI Services

-
 Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award
Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award
-
US renews offer of $100 mn to Cuba if it cooperates

-
 City still 'alive' but need Arsenal slip: Guardiola
City still 'alive' but need Arsenal slip: Guardiola
-
Man City ease past Palace to keep pressure on Arsenal

-
 Alaves end champions Barca's bid for 100-point record
Alaves end champions Barca's bid for 100-point record
-
US jury begins deliberations on 737 MAX victim suit against Boeing

-
 PSG clinch fifth straight Ligue 1 title
PSG clinch fifth straight Ligue 1 title
-
Inter Milan win Italian Cup to secure domestic double

-
 Man City see off Palace to keep pressure on Arsenal
Man City see off Palace to keep pressure on Arsenal
-
Trump and Xi set for high-stakes talks in Beijing

-
 S&P 500, Nasdaq end at records as oil prices retreat
S&P 500, Nasdaq end at records as oil prices retreat
-
Iran holds World Cup send-off for national football team

-
 McIlroy's toe 'totally fine' after nine-hole PGA practice
McIlroy's toe 'totally fine' after nine-hole PGA practice
-
Rare 'Ocean Dream' blue-green diamond sells for $17 mn at auction

-
 California says probing possible violations over World Cup ticket sales
California says probing possible violations over World Cup ticket sales
-
US races to secure rare earths to rebuild depleted arsenal

-
 Matthew Perry drug middleman jailed for two years
Matthew Perry drug middleman jailed for two years
-
Warsh confirmed as Fed chair as central bank faces Trump assault


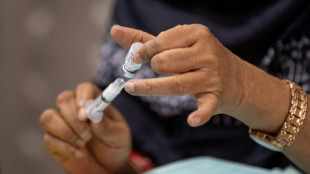
Japan approves stem-cell treatment for Parkinson's in world first
Japan has approved ground-breaking stem-cell treatments for Parkinson's and severe heart failure, one of the manufacturers and media reports said Friday, with the therapies expected to reach patients within months.
Pharmaceutical company Sumitomo Pharma said it received the green light for the manufacture and sale of Amchepry, its Parkinson's disease treatment that transplants stem cells into a patient's brain.
Japan's health ministry also gave the go-ahead to ReHeart, heart muscle sheets developed by medical startup Cuorips that can help form new blood vessels and restore heart function, media reports said.
The treatments could be on the market and rolled out to patients as early as this summer, reports said, citing the health ministry, becoming the world's first commercially available medical products using (iPS) cells.
Japanese scientist Shinya Yamanaka won the Nobel Prize in 2012 for his research into iPS, which have the potential to develop into any cell in the body.
"I hope this will bring relief to patients not only in Japan but around the world," health minister Kenichiro Ueno told a press conference.
"We will promptly carry out all necessary procedures to ensure it reaches all patients without fail."
In a statement, Sumitomo Pharma said it had obtained "conditional and time-limited approval" for the manufacture and marketing of Amchepry under a system which is reportedly designed to get these products to patients as quickly as possible.
The approval is a kind of "provisional license", the Asahi newspaper said, after the safety and efficacy of the treatment was judged based on data from fewer patients than in ordinary clinical trials for drugs.
A trial led by Kyoto University researchers indicated that the company's treatment was safe and successful in improving symptoms.
The study involved seven Parkinson's patients aged between 50 and 69, with each receiving a total of either five million or 10 million cells implanted on both sides of the brain.
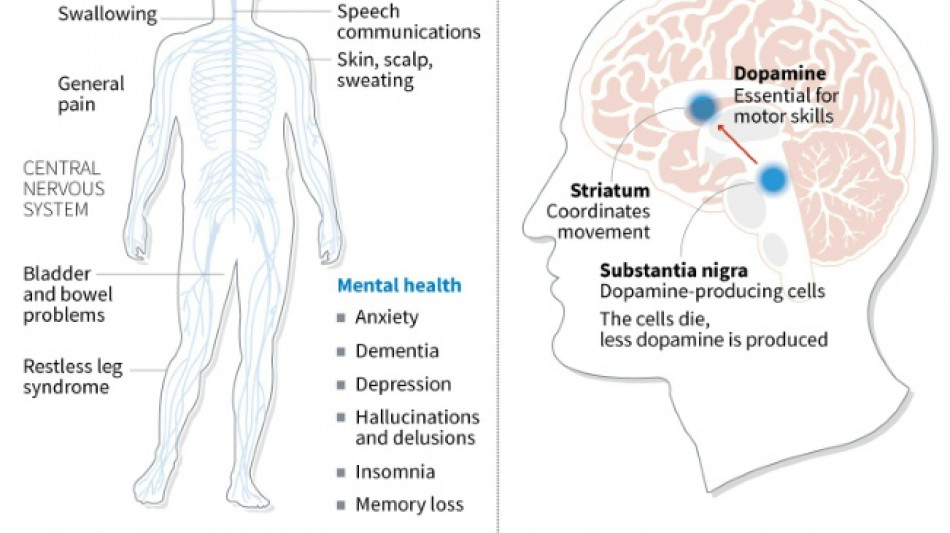
The iPS cells from healthy donors were developed into the precursors of dopamine-producing brain cells, which are no longer present in people with Parkinson's disease.
The patients were monitored for two years and no major adverse effects were found, the study said. Four patients showed improvements in symptoms.
Parkinson's disease is a chronic, degenerative neurological disorder that affects the body's motor system, often causing shaking and other difficulties in movement.
Worldwide, about 10 million people have the illness, according to the Parkinson's Foundation.
Currently available therapies "improve symptoms without slowing or halting the disease progression," the foundation says.
iPS cells are created by stimulating mature, already specialised, cells back into a juvenile state -- basically cloning without the need for an embryo.
The cells can be transformed into a range of different types of cells, and their use is a key sector of medical research.
O.Brown--AT