-
 Denmark, Australia in the spotlight in Eurovision second semi
Denmark, Australia in the spotlight in Eurovision second semi
-
Heavy Russian strikes on Kyiv kill one, wound 31

-
 Xi warns Trump on Taiwan at Beijing summit
Xi warns Trump on Taiwan at Beijing summit
-
Iran war and oil dominate BRICS meet in India

-
 Bone appetit: Paris pups lap up treats at dog-centric spots
Bone appetit: Paris pups lap up treats at dog-centric spots
-
Kohli senses end after roaring back to form with IPL century

-
 India bars sugar exports until September
India bars sugar exports until September
-
Madonna, Shakira, BTS to headline first World Cup final half-time show

-
 Japan takes 'half step' toward fixing slow retrial system
Japan takes 'half step' toward fixing slow retrial system
-
Honda posts operating loss, first since 1957

-
 Madonna, Shakira, BTS to headline World Cup final half-time show
Madonna, Shakira, BTS to headline World Cup final half-time show
-
A quarter of World Cup games risk searing heat: scientists

-
 Six hantavirus cruise passengers head to Australia
Six hantavirus cruise passengers head to Australia
-
Suspect detained in Philippine senate gunfire: police

-
 Cavs top Pistons in overtime for 3-2 series lead
Cavs top Pistons in overtime for 3-2 series lead
-
Canadian football ready for World Cup coming out party

-
 US court suspends sanctions on UN expert on Palestinians
US court suspends sanctions on UN expert on Palestinians
-
Asia markets mixed as Trump-Xi summit, AI trade dominate

-
 'Promised to us': The Israelis dreaming of settling south Lebanon
'Promised to us': The Israelis dreaming of settling south Lebanon
-
'Rare, meaningful': North Korean football team ventures into South

-
 In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
-
Historic Swiss solar-powered plane crashes into sea

-
 A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
-
Indian pharma fuels Africa's 'zombie drug' and opioid crisis

-
 After months of blackout, Iran gives internet to select few
After months of blackout, Iran gives internet to select few
-
Wood urges New Zealand to 'create some history' at World Cup

-
 In Washington, the fight to preserve Black cemeteries
In Washington, the fight to preserve Black cemeteries
-
US children's book author sentenced to life after poisoning husband

-
 Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
-
Battery X Metals Achieves Milestone with Delivery of Next-Generation Patent-Pending Lithium-Ion Battery Rebalancing Machine Featuring Design Enhancements, Advancing Strategic Commercialization Initiatives

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14
InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14
-
Akkodis Recognized in HFS Horizons 2026 Report for Enterprise Ready Agentic AI Services

-
 Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award
Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award
-
US renews offer of $100 mn to Cuba if it cooperates

-
 City still 'alive' but need Arsenal slip: Guardiola
City still 'alive' but need Arsenal slip: Guardiola
-
Man City ease past Palace to keep pressure on Arsenal

-
 Alaves end champions Barca's bid for 100-point record
Alaves end champions Barca's bid for 100-point record
-
US jury begins deliberations on 737 MAX victim suit against Boeing

-
 PSG clinch fifth straight Ligue 1 title
PSG clinch fifth straight Ligue 1 title
-
Inter Milan win Italian Cup to secure domestic double

-
 Man City see off Palace to keep pressure on Arsenal
Man City see off Palace to keep pressure on Arsenal
-
Trump and Xi set for high-stakes talks in Beijing

-
 S&P 500, Nasdaq end at records as oil prices retreat
S&P 500, Nasdaq end at records as oil prices retreat
-
Iran holds World Cup send-off for national football team

-
 McIlroy's toe 'totally fine' after nine-hole PGA practice
McIlroy's toe 'totally fine' after nine-hole PGA practice
-
Rare 'Ocean Dream' blue-green diamond sells for $17 mn at auction

-
 California says probing possible violations over World Cup ticket sales
California says probing possible violations over World Cup ticket sales
-
US races to secure rare earths to rebuild depleted arsenal

-
 Matthew Perry drug middleman jailed for two years
Matthew Perry drug middleman jailed for two years
-
Warsh confirmed as Fed chair as central bank faces Trump assault


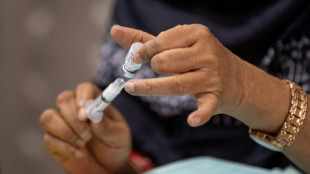
US to remove warnings from menopause hormone therapy
The US health regulatory agency on Monday said it would direct manufacturers to remove a bold warning on many hormone therapies used to alleviate menopausal symptoms, saying the risks have been exaggerated.
Hormone Replacement Therapy can be taken to replace estrogen that the body stops producing during menopause -- the natural process that ends female reproductive years -- with the aim of alleviating symptoms that can be physically and mentally debilitating including hot flashes, brain fog, insomnia, night sweats, joint pain and bone loss.
It once was used routinely, but a major 2002 study that was aimed at exploring how the therapies could prevent chronic disease pointed to risks associated with specific HRT formulations.
Since then "black box warnings" -- the strongest warning the US Food and Drug Administration can require on prescription drugs -- have sounded alarm over increased HRT risks including of certain cancers, cardiovascular conditions and probable dementia.
Prescription and use of the therapies over recent decades plummeted.
But the matter remains one of debate, and critics have pointed to flaws with the early-2000s study, whose trials were halted as risks appeared: namely it focused on women who were on average a decade-post menopause and in their 60s, when cardiovascular risks increase regardless.
Today guidance generally indicates that newly menopausal or perimenopausal women -- broadly in their late 40s or 50s -- are potential candidates for treatment.
There also are newer, more localized or lower-dose forms of the therapies available.
"We're challenging outdated thinking and recommitting to evidence-based medicine that empowers rather than restricts," US health chief Robert F. Kennedy Jr said in introducing the measure.
- More nuance -
Many members of the medical community have urged revisiting the label, which they say can scare women for whom benefits may outweigh risks.
Others have voiced concern that changes shouldn't come without a transparent review process that convenes independent experts.
FDA head Marty Makary dismissed that notion in a Monday briefing, saying such committees are "bureaucratic, long, often conflicted and very expensive.
"People have felt demoralized in this field. We've made almost no progress in assessing the evidence for years."
FDA officials have emphasized the new label will provide for more nuanced discussion between patients and doctors in evaluating whether HRT can be right for individuals.
Typically women in a low-risk category have healthy vitals including weight and blood pressure as well as normal-range cholesterol levels, with no history or a lower risk of developing breast cancer. They are also within the first 10 years of beginning menopause.
The president of the American College of Obstetricians and Gynecologists, Steven Fleischman, commended the FDA's move, saying "the updated labels will better allow patients and clinicians to engage in a shared decision-making process."
Fleischman said in a statement the change would not shift the ACOG's guidance on estrogen therapy, but noted it was important to distinguish between systemic estrogen products -- including methods taken orally or via patches -- and low-dose vaginal estrogen.
"Like all medications, systemic estrogen products are not without risk, and their use should be based on an individualized conversation between patients and their clinicians," the health association said.
The FDA said it is not seeking to remove the boxed warning for endometrial cancer for systemic estrogen-alone products.
O.Gutierrez--AT