-
 Russia pummels Kyiv, killing three and denting peace hopes
Russia pummels Kyiv, killing three and denting peace hopes
-
Subdued Trump left waiting for 'big hug' from Xi

-
 Slot has 'every reason to believe' he will remain as Liverpool boss
Slot has 'every reason to believe' he will remain as Liverpool boss
-
British PM battles to stay in power amid rebellion

-
 Ex-Philippine drug war enforcer flees Senate refuge
Ex-Philippine drug war enforcer flees Senate refuge
-
U2 surprise fans in Mexico City to shoot music video

-
 Asia stocks uneven as investors assess high-stakes Trump-Xi talks, AI rally
Asia stocks uneven as investors assess high-stakes Trump-Xi talks, AI rally
-
Burberry returns to full-year profit on turnaround plan

-
 Spiky, polarising, rarely dull: ups and downs of rugby's Eddie Jones
Spiky, polarising, rarely dull: ups and downs of rugby's Eddie Jones
-
Denmark, Australia in the spotlight in Eurovision second semi

-
 Heavy Russian strikes on Kyiv kill one, wound 31
Heavy Russian strikes on Kyiv kill one, wound 31
-
Xi warns Trump on Taiwan at Beijing summit

-
 Iran war and oil dominate BRICS meet in India
Iran war and oil dominate BRICS meet in India
-
Bone appetit: Paris pups lap up treats at dog-centric spots

-
 Kohli senses end after roaring back to form with IPL century
Kohli senses end after roaring back to form with IPL century
-
India bars sugar exports until September

-
 Madonna, Shakira, BTS to headline first World Cup final half-time show
Madonna, Shakira, BTS to headline first World Cup final half-time show
-
Japan takes 'half step' toward fixing slow retrial system

-
 Honda posts operating loss, first since 1957
Honda posts operating loss, first since 1957
-
Madonna, Shakira, BTS to headline World Cup final half-time show

-
 A quarter of World Cup games risk searing heat: scientists
A quarter of World Cup games risk searing heat: scientists
-
Six hantavirus cruise passengers head to Australia

-
 Suspect detained in Philippine senate gunfire: police
Suspect detained in Philippine senate gunfire: police
-
Cavs top Pistons in overtime for 3-2 series lead

-
 Canadian football ready for World Cup coming out party
Canadian football ready for World Cup coming out party
-
US court suspends sanctions on UN expert on Palestinians

-
 Asia markets mixed as Trump-Xi summit, AI trade dominate
Asia markets mixed as Trump-Xi summit, AI trade dominate
-
'Promised to us': The Israelis dreaming of settling south Lebanon

-
 'Rare, meaningful': North Korean football team ventures into South
'Rare, meaningful': North Korean football team ventures into South
-
In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS

-
 Historic Swiss solar-powered plane crashes into sea
Historic Swiss solar-powered plane crashes into sea
-
A woman UN leader is 'historical justice,' says Ecuadoran contender for top job

-
 Indian pharma fuels Africa's 'zombie drug' and opioid crisis
Indian pharma fuels Africa's 'zombie drug' and opioid crisis
-
After months of blackout, Iran gives internet to select few

-
 Wood urges New Zealand to 'create some history' at World Cup
Wood urges New Zealand to 'create some history' at World Cup
-
In Washington, the fight to preserve Black cemeteries

-
 US children's book author sentenced to life after poisoning husband
US children's book author sentenced to life after poisoning husband
-
Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes

-
 NAV Fund Services (Ireland) Ltd. Expands European Fund Servicing Offering to Support UCITS Funds
NAV Fund Services (Ireland) Ltd. Expands European Fund Servicing Offering to Support UCITS Funds
-
AVI Urges the Dismissal of Two Directors at Wacom

-
 PLAS-LABS Simplifies Scientific Validation With Automated Citation Tracking Powered by Bioz
PLAS-LABS Simplifies Scientific Validation With Automated Citation Tracking Powered by Bioz
-
Battery X Metals Achieves Milestone with Delivery of Next-Generation Patent-Pending Lithium-Ion Battery Rebalancing Machine Featuring Design Enhancements, Advancing Strategic Commercialization Initiatives

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14
InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14
-
Akkodis Recognized in HFS Horizons 2026 Report for Enterprise Ready Agentic AI Services

-
 Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award
Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award
-
US renews offer of $100 mn to Cuba if it cooperates

-
 City still 'alive' but need Arsenal slip: Guardiola
City still 'alive' but need Arsenal slip: Guardiola
-
Man City ease past Palace to keep pressure on Arsenal

-
 Alaves end champions Barca's bid for 100-point record
Alaves end champions Barca's bid for 100-point record
-
US jury begins deliberations on 737 MAX victim suit against Boeing


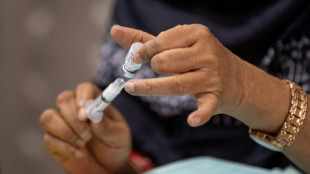
US to limit Covid boosters to over-65s or those at high risk
The United States will limit routine Covid-19 boosters to people over 65 or those at higher risk of serious illness, while requiring new placebo-controlled trials to justify vaccines for healthy individuals under that threshold, senior health officials said Tuesday.
In a letter to the prestigious New England Journal of Medicine, the Food and Drug Administration's Vinayak Prasad and Commissioner Martin Makary said the shift brought US policy more in line with European nations.
They described the initial rollout of Covid-19 vaccines as "a major scientific, medical, and regulatory accomplishment" -- but argued that the benefits of repeated boosters in low-risk individuals remained uncertain.
They contrasted the US approach with that of countries such as the United Kingdom, France and Germany, which limit booster recommendations to older adults and those with underlying conditions.
Going forward, the FDA believes it will continue to find the benefit-risk balance favorable for people over 65, and people over the age of six months with one or more underlying conditions.
However, "for all healthy persons -- those with no risk factors for severe Covid-19 -- between the ages of 6 months and 64 years, the FDA anticipates the need for randomized, controlled trial data," they said.
"The control group could receive a saline placebo."
They identified adults aged 50 to 64 as an ideal study population, and said trials should assess whether boosters reduce symptomatic illness, severe disease, hospitalization, and death.
Makary and Prasad also sought to reassure those worried about losing access to vaccines under the new framework.
The Centers for Disease Control and Prevention's definition of risk factors is "vast, including obesity and even mental health conditions such as depression," they wrote, adding that between 100 million and 200 million Americans would likely still qualify under this guidance.
Prasad, a hematologist-oncologist who now leads the FDA's Center for Biologics Evaluation and Research (CBER), rose to prominence during the pandemic for publicly questioning the widespread use of boosters.
M.O.Allen--AT