-
 Burberry returns to full-year profit on turnaround plan
Burberry returns to full-year profit on turnaround plan
-
Spiky, polarising, rarely dull: ups and downs of rugby's Eddie Jones

-
 Denmark, Australia in the spotlight in Eurovision second semi
Denmark, Australia in the spotlight in Eurovision second semi
-
Heavy Russian strikes on Kyiv kill one, wound 31

-
 Xi warns Trump on Taiwan at Beijing summit
Xi warns Trump on Taiwan at Beijing summit
-
Iran war and oil dominate BRICS meet in India

-
 Bone appetit: Paris pups lap up treats at dog-centric spots
Bone appetit: Paris pups lap up treats at dog-centric spots
-
Kohli senses end after roaring back to form with IPL century

-
 India bars sugar exports until September
India bars sugar exports until September
-
Madonna, Shakira, BTS to headline first World Cup final half-time show

-
 Japan takes 'half step' toward fixing slow retrial system
Japan takes 'half step' toward fixing slow retrial system
-
Honda posts operating loss, first since 1957

-
 Madonna, Shakira, BTS to headline World Cup final half-time show
Madonna, Shakira, BTS to headline World Cup final half-time show
-
A quarter of World Cup games risk searing heat: scientists

-
 Six hantavirus cruise passengers head to Australia
Six hantavirus cruise passengers head to Australia
-
Suspect detained in Philippine senate gunfire: police

-
 Cavs top Pistons in overtime for 3-2 series lead
Cavs top Pistons in overtime for 3-2 series lead
-
Canadian football ready for World Cup coming out party

-
 US court suspends sanctions on UN expert on Palestinians
US court suspends sanctions on UN expert on Palestinians
-
Asia markets mixed as Trump-Xi summit, AI trade dominate

-
 'Promised to us': The Israelis dreaming of settling south Lebanon
'Promised to us': The Israelis dreaming of settling south Lebanon
-
'Rare, meaningful': North Korean football team ventures into South

-
 In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
-
Historic Swiss solar-powered plane crashes into sea

-
 A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
-
Indian pharma fuels Africa's 'zombie drug' and opioid crisis

-
 After months of blackout, Iran gives internet to select few
After months of blackout, Iran gives internet to select few
-
Wood urges New Zealand to 'create some history' at World Cup

-
 In Washington, the fight to preserve Black cemeteries
In Washington, the fight to preserve Black cemeteries
-
US children's book author sentenced to life after poisoning husband

-
 Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
-
PLAS-LABS Simplifies Scientific Validation With Automated Citation Tracking Powered by Bioz

-
 Battery X Metals Achieves Milestone with Delivery of Next-Generation Patent-Pending Lithium-Ion Battery Rebalancing Machine Featuring Design Enhancements, Advancing Strategic Commercialization Initiatives
Battery X Metals Achieves Milestone with Delivery of Next-Generation Patent-Pending Lithium-Ion Battery Rebalancing Machine Featuring Design Enhancements, Advancing Strategic Commercialization Initiatives
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14

-
 Akkodis Recognized in HFS Horizons 2026 Report for Enterprise Ready Agentic AI Services
Akkodis Recognized in HFS Horizons 2026 Report for Enterprise Ready Agentic AI Services
-
Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award

-
 US renews offer of $100 mn to Cuba if it cooperates
US renews offer of $100 mn to Cuba if it cooperates
-
City still 'alive' but need Arsenal slip: Guardiola

-
 Man City ease past Palace to keep pressure on Arsenal
Man City ease past Palace to keep pressure on Arsenal
-
Alaves end champions Barca's bid for 100-point record
-
 US jury begins deliberations on 737 MAX victim suit against Boeing
US jury begins deliberations on 737 MAX victim suit against Boeing
-
PSG clinch fifth straight Ligue 1 title

-
 Inter Milan win Italian Cup to secure domestic double
Inter Milan win Italian Cup to secure domestic double
-
Man City see off Palace to keep pressure on Arsenal

-
 Trump and Xi set for high-stakes talks in Beijing
Trump and Xi set for high-stakes talks in Beijing
-
S&P 500, Nasdaq end at records as oil prices retreat

-
 Iran holds World Cup send-off for national football team
Iran holds World Cup send-off for national football team
-
McIlroy's toe 'totally fine' after nine-hole PGA practice

-
 Rare 'Ocean Dream' blue-green diamond sells for $17 mn at auction
Rare 'Ocean Dream' blue-green diamond sells for $17 mn at auction
-
California says probing possible violations over World Cup ticket sales


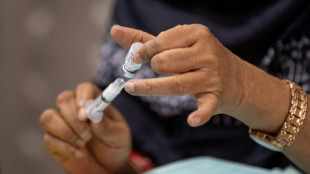
Moderna Covid vaccine gets full US approval
The US Food and Drug Administration (FDA) announced Monday that it has granted full approval to Moderna's Covid-19 "Spikevax" vaccine, which had previously received an emergency use authorization in the United States.
"The public can be assured that Spikevax meets the FDA's high standards for safety, effectiveness and manufacturing quality required of any vaccine approved for use in the United States," acting FDA commissioner Janet Woodcock said in a statement.
"The FDA's approval of Spikevax is a significant step in the fight against the Covid-19 pandemic, marking the second vaccine approved to prevent Covid-19," Woodcock said.
The full approval of the Moderna vaccine is for individuals aged 18 or older.
A Pfizer vaccine for individuals aged 16 or older received full FDA approval at the end of August.
Woodcock said she hoped FDA approval of the Moderna vaccine "may instill additional confidence in making the decision to get vaccinated."
Moderna CEO Stephane Bancel called the FDA move a "momentous milestone" for the company.
"Our Covid-19 vaccine has been administered to hundreds of millions of people around the world, protecting people from Covid-19 infection, hospitalization and death," Bancel said in a statement.
The Moderna vaccine received an emergency use authorization in December 2020.
The full approval from the FDA concerns the first two doses of the Moderna vaccine. A booster dose remains under an emergency use authorization.
The FDA said clinical trials to determine the effectiveness of Spikevax included 14,287 vaccine recipients and 14,164 placebo recipients.
The most common side effects by clinical trial participants were pain at the injection site, fatigue, headaches, muscle or joint pain, chills, nausea or vomiting and swollen lymph nodes, the FDA said.
It said there was an increased risk in men aged 18 to 24 of myocarditis -- inflammation of the heart muscle -- and pericarditis -- inflammation of tissue surrounding the heart -- following vaccination with the second dose.
But, the FDA said, it has determined that the "benefits of the vaccine outweigh the risk of myocarditis and pericarditis in individuals 18 years of age and older."
M.King--AT