
-
 Russian tanker set to deliver oil to crisis-hit Cuba
Russian tanker set to deliver oil to crisis-hit Cuba
-
Iran fires missiles across Middle East as Trump threatens oil hub

-
 Indonesia summons Google, Meta for 'not complying' with teen social media ban: minister
Indonesia summons Google, Meta for 'not complying' with teen social media ban: minister
-
Wembanyama at the double as Spurs beat Bulls

-
 Australia investigates tech giants over social media ban breaches
Australia investigates tech giants over social media ban breaches
-
Hindu devotional clubbing sways India's youth

-
 Oil slips, stocks rise as report says Trump willing to end war
Oil slips, stocks rise as report says Trump willing to end war
-
Mind games: How football stars are fuelling chess boom

-
 Indonesia trims meals programme: what next?
Indonesia trims meals programme: what next?
-
'A very big deal': Canadian astronaut reflects on historic Moon mission

-
 US pro table tennis league blasts niche sport into spotlight
US pro table tennis league blasts niche sport into spotlight
-
Iran defiant as Trump threatens to destroy oil island

-
 Trump threatens to destroy Iran oil island despite claims of talks
Trump threatens to destroy Iran oil island despite claims of talks
-
NASA begins countdown to April 1 Moon launch

-
 NBA Bulls fire Ivey after anti-LGBTQ comments
NBA Bulls fire Ivey after anti-LGBTQ comments
-
Australian regulator probes Facebook, YouTube over teen social media ban

-
 Iraq coach shielding players from war ahead of World Cup bid
Iraq coach shielding players from war ahead of World Cup bid
-
Undav rescues Germany late in Ghana friendly

-
 Messi to start for Argentina in World Cup send-off: Scaloni
Messi to start for Argentina in World Cup send-off: Scaloni
-
Oil rises on Trump's Iran threats, stocks mixed

-
 After pope's remark, White House defends praying for US troops
After pope's remark, White House defends praying for US troops
-
Powell probe leaves US Fed leadership change in limbo

-
 Celine Dion announces comeback following health struggle
Celine Dion announces comeback following health struggle
-
'Is it Kafka?' US judge baffled by new Pentagon press policy

-
 Cubans ready for Russian oil but some say not enough
Cubans ready for Russian oil but some say not enough
-
Teen Suryavanshi shines as Rajasthan hammer Chennai in IPL

-
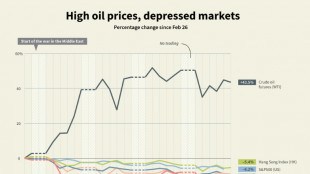 Stock market winners and losers one month into US-Israel war on Iran
Stock market winners and losers one month into US-Israel war on Iran
-
Hodgson says surprise return to management is only for short-term

-
 What could Trump achieve by threatening Iran's Kharg Island?
What could Trump achieve by threatening Iran's Kharg Island?
-
India declares victory over Maoist insurgency

-
 Germany's Merz pushes return of Syrians as he hosts leader Sharaa
Germany's Merz pushes return of Syrians as he hosts leader Sharaa
-
G7 ministers pledge 'necessary measures' to ensure stable energy market

-
 Cardiff City lose compensation case over Emiliano Sala death
Cardiff City lose compensation case over Emiliano Sala death
-
Several French far-right mayors take down EU flags

-
 Air Canada CEO to retire after row over English-only condolence message
Air Canada CEO to retire after row over English-only condolence message
-
Oil rises on Trump's Iran threats, stocks take cue on talks

-
 Syrian leader pledges to work with Germany on migration, recovery
Syrian leader pledges to work with Germany on migration, recovery
-
AI agent future is coming, OpenClaw creator tells AFP

-
 Cardiff lose 122 mn euro compensation case over Emiliano Sala death
Cardiff lose 122 mn euro compensation case over Emiliano Sala death
-
Tuchel defends Rice and Saka after England withdrawals

-
 G7 ministers tackle economic fallout of Mideast war
G7 ministers tackle economic fallout of Mideast war
-
Tottenham close in on De Zerbi as next boss - reports

-
 Kenya's former NY marathon champion Korir gets 5-year doping ban
Kenya's former NY marathon champion Korir gets 5-year doping ban
-
Lukaku says 'could never turn back on Napoli' after treatment row

-
 Syrian leader visits Germany to talk war, recovery, refugees
Syrian leader visits Germany to talk war, recovery, refugees
-
Renault says developing ground-based military drone

-
 Iran hangs two 'political prisoners' from banned opposition: activists
Iran hangs two 'political prisoners' from banned opposition: activists
-
Russia expels UK diplomat on spying allegations

-
 Premier League fans back call to scrap VAR
Premier League fans back call to scrap VAR
-
Italy hoping to scale World Cup 'Everest' ahead of Bosnia play-off showdown


Biogen pulls controversial Alzheimer's drug Aduhelm
A controversial Alzheimer's drug that was trumpeted as the first to ever treat the cognitive decline associated with the devastating brain disorder has been pulled from the market, its maker Biogen announced Wednesday.
The US Food and Drug Administration awarded accelerated approval to Aduhelm in June 2021, a decision that was highly contentious at the time because the agency overruled its own independent advisors, who found there was insufficient evidence of benefit.
At least three of the 11-member independent committee that voted unanimously against recommending the drug subsequently resigned, and US congressional investigators slammed the accelerated approval as "rife with irregularities."
Biogen said it was discontinuing Aduhelm to put more resources into Leqembi, a newer Alzheimer's medicine that was fully approved last year under the traditional regulatory pathway.
"When searching for new medicines, one breakthrough can be the foundation that triggers future medicines to be developed," said Christopher Viehbacher, president and CEO of the Cambridge, Massachusetts-based biotech firm.
"Aduhelm was that groundbreaking discovery that paved the way for a new class of drugs and reinvigorated investments in the field."
Aduhelm, a monoclonal antibody that targets the build-up of a protein called amyloid beta in the brain tissue which is thought to be a cause of Alzheimer's, was tested in two late-stage human trials.
It showed a reduction in cognitive decline in one of the studies, but not the other.
According to a congressional report from December 2022, the FDA "considered Aduhelm under the traditional approval pathway used for most drugs for nine months, before abruptly changing course and granting approval under the accelerated approval pathway after a three-week review period."
The report said that FDA interactions with Biogen were "atypical" and included a failure to properly document contacts between agency staff and the drug maker.
The FDA and Biogen had also "inappropriately collaborated" on a joint briefing document for a key advisory committee, it said.
"FDA's approval process was rife with irregularities."
As for Biogen, the report said the company "viewed Aduhelm as an unprecedented financial opportunity -- estimating a potential peak revenue of $18 billion per year."
The congressional panel pointed to an "unjustifiably high price" for Aduhelm of $56,000 a year for patients.
Biogen's Leqembi, which it co-manufactures with Eisai of Japan, is now the only US approved treatment for Alzheimer's. It also targets amyloid beta and has been found to modestly reduce cognitive decline in patients with early stage disease.
Donanemab, developed by Eli Lilly, could be next to get the green light after performing similarly in clinical trials.
Alzheimer's is the most common form of dementia. More than one in nine people over 65 develop the condition, which worsens over time, robbing them of their memories and independence, according to the US Alzheimer's Association.
H.Gonzales--AT




