-
 U2 surprise fans in Mexico City to shoot music video
U2 surprise fans in Mexico City to shoot music video
-
Asia stocks uneven as investors assess high-stakes Trump-Xi talks, AI rally

-
 Burberry returns to full-year profit on turnaround plan
Burberry returns to full-year profit on turnaround plan
-
Spiky, polarising, rarely dull: ups and downs of rugby's Eddie Jones

-
 Denmark, Australia in the spotlight in Eurovision second semi
Denmark, Australia in the spotlight in Eurovision second semi
-
Heavy Russian strikes on Kyiv kill one, wound 31

-
 Xi warns Trump on Taiwan at Beijing summit
Xi warns Trump on Taiwan at Beijing summit
-
Iran war and oil dominate BRICS meet in India

-
 Bone appetit: Paris pups lap up treats at dog-centric spots
Bone appetit: Paris pups lap up treats at dog-centric spots
-
Kohli senses end after roaring back to form with IPL century

-
 India bars sugar exports until September
India bars sugar exports until September
-
Madonna, Shakira, BTS to headline first World Cup final half-time show

-
 Japan takes 'half step' toward fixing slow retrial system
Japan takes 'half step' toward fixing slow retrial system
-
Honda posts operating loss, first since 1957

-
 Madonna, Shakira, BTS to headline World Cup final half-time show
Madonna, Shakira, BTS to headline World Cup final half-time show
-
A quarter of World Cup games risk searing heat: scientists

-
 Six hantavirus cruise passengers head to Australia
Six hantavirus cruise passengers head to Australia
-
Suspect detained in Philippine senate gunfire: police

-
 Cavs top Pistons in overtime for 3-2 series lead
Cavs top Pistons in overtime for 3-2 series lead
-
Canadian football ready for World Cup coming out party

-
 US court suspends sanctions on UN expert on Palestinians
US court suspends sanctions on UN expert on Palestinians
-
Asia markets mixed as Trump-Xi summit, AI trade dominate

-
 'Promised to us': The Israelis dreaming of settling south Lebanon
'Promised to us': The Israelis dreaming of settling south Lebanon
-
'Rare, meaningful': North Korean football team ventures into South

-
 In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
In-form Messi hits brace as Miami win 5-3 at Cincinnati in MLS
-
Historic Swiss solar-powered plane crashes into sea

-
 A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
A woman UN leader is 'historical justice,' says Ecuadoran contender for top job
-
Indian pharma fuels Africa's 'zombie drug' and opioid crisis

-
 After months of blackout, Iran gives internet to select few
After months of blackout, Iran gives internet to select few
-
Wood urges New Zealand to 'create some history' at World Cup

-
 In Washington, the fight to preserve Black cemeteries
In Washington, the fight to preserve Black cemeteries
-
US children's book author sentenced to life after poisoning husband

-
 Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
Emotional Vin Diesel leads 'Fast and Furious' tribute in Cannes
-
AVI Urges the Dismissal of Two Directors at Wacom

-
 PLAS-LABS Simplifies Scientific Validation With Automated Citation Tracking Powered by Bioz
PLAS-LABS Simplifies Scientific Validation With Automated Citation Tracking Powered by Bioz
-
Battery X Metals Achieves Milestone with Delivery of Next-Generation Patent-Pending Lithium-Ion Battery Rebalancing Machine Featuring Design Enhancements, Advancing Strategic Commercialization Initiatives

-
 InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14
InterContinental Hotels Group PLC Announces Transaction in Own Shares - May 14
-
Akkodis Recognized in HFS Horizons 2026 Report for Enterprise Ready Agentic AI Services

-
 Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award
Lexus Of Oakville Recognized for Redefining the Luxury Dealership Experience With 2026 Consumer Choice Award
-
US renews offer of $100 mn to Cuba if it cooperates

-
 City still 'alive' but need Arsenal slip: Guardiola
City still 'alive' but need Arsenal slip: Guardiola
-
Man City ease past Palace to keep pressure on Arsenal

-
 Alaves end champions Barca's bid for 100-point record
Alaves end champions Barca's bid for 100-point record
-
US jury begins deliberations on 737 MAX victim suit against Boeing

-
 PSG clinch fifth straight Ligue 1 title
PSG clinch fifth straight Ligue 1 title
-
Inter Milan win Italian Cup to secure domestic double

-
 Man City see off Palace to keep pressure on Arsenal
Man City see off Palace to keep pressure on Arsenal
-
Trump and Xi set for high-stakes talks in Beijing

-
 S&P 500, Nasdaq end at records as oil prices retreat
S&P 500, Nasdaq end at records as oil prices retreat
-
Iran holds World Cup send-off for national football team


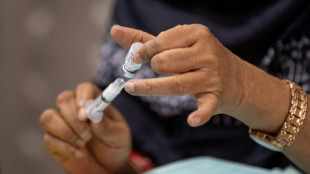
US authorizes first condom for use in anal sex
The US Food and Drug Administration (FDA) on Wednesday authorized the first condom for use during anal intercourse, in what was hailed as a victory for sexual health by experts.
Although people already use condoms for anal sex -- as is recommended by health agencies including the Centers for Disease Control and Prevention (CDC) -- regulators across the world had only previously allowed companies to officially market their products as "safe and effective" for vaginal use.
Sexual health advocates considered this an unmet public health need since unprotected anal intercourse carries the greatest risk of HIV transmission via sexual exposure, with one study finding that 69 percent of men who have sex with men would use condoms more frequently if they were FDA-indicated.
Wednesday's authorization of Global Protection Corp's One Male Condom follows a clinical trial involving more than 500 people, carried out by Emory University.
"The FDA's authorization of a condom that is specifically indicated, evaluated and labeled for anal intercourse may improve the likelihood of condom use during anal intercourse," said agency scientist Courtney Lias in a statement.
The condom is also indicated to prevent sexually transmitted infections -- and as a contraceptive -- during vaginal sex.
"We want people to have lots of sex -- but we also want them to be empowered and informed," said Davin Wedel, president of Global Protection Corp, which makes the condom brand that is available in 54 sizes, and incorporates a paper template to help each user find the right size.
The clinical trial involved 252 men who have sex with men and 252 men who have sex with women, aged between 18 and 54.
The FDA had said it would accept a five percent failure rate, which previous trials had failed to accomplish. The limit was easily surpassed in the new study, with the failure rate 0.68 percent for anal and 1.89 percent for vaginal intercourse.
The researchers behind the study, which was published in The Lancet's eClinicalMedicine, said one of the reasons the trial succeeded where others failed in the past was likely due to the provision of lubricant and inclusion of instructions on how to use the product.
Lubricant reduces friction, which in turn causes condom failure from slippage and breakage.
Another reason could be that participants were asked to keep mobile phone-based daily diaries, whereas past trials had asked volunteers to recall failure events up to several months later.
Monica Gandhi, an infectious disease doctor and medical director of an HIV clinic in San Francisco, welcomed the finding.
"The important thing about condoms is they don't just prevent HIV, but they prevent gonorrhea, chlamydia and syphilis," she told AFP, adding it was surprising that such an authorization had taken so long to achieve.
In its statement, the FDA said the green light could pave the way for more makers to apply for similar authorization if they show equivalent results.
T.Sanchez--AT