
-
 Deposed and detained: Niger president's fate unclear nearly three years on
Deposed and detained: Niger president's fate unclear nearly three years on
-
Newcastle say no manager change 'at the moment'

-
 Newly-hatched rare Indian bustard chick gets 50-strong guard
Newly-hatched rare Indian bustard chick gets 50-strong guard
-
Stranded whale frees itself again off German coast

-
 Archaeologists forced by Mideast war to cut short Iraq digs
Archaeologists forced by Mideast war to cut short Iraq digs
-
Stranded whale frees itself again off German coast and disappears

-
 Thailand's king endorses new cabinet
Thailand's king endorses new cabinet
-
China bans entombing cremated remains in empty flats

-
 Calls grow for 15-year-old Suryavanshi to make India bow
Calls grow for 15-year-old Suryavanshi to make India bow
-
Stocks slip, oil swings after report says Trump willing to end war

-
 Pakistan cricketer Naseem fined record $71,500 for minister criticism
Pakistan cricketer Naseem fined record $71,500 for minister criticism
-
China teen diving prodigy nearly retired after 'reaching mental limit'

-
 Myanmar junta chief elected vice-president
Myanmar junta chief elected vice-president
-
Russian tanker set to deliver oil to crisis-hit Cuba

-
 Iran fires missiles across Middle East as Trump threatens oil hub
Iran fires missiles across Middle East as Trump threatens oil hub
-
Indonesia summons Google, Meta for 'not complying' with teen social media ban: minister

-
 Wembanyama at the double as Spurs beat Bulls
Wembanyama at the double as Spurs beat Bulls
-
Australia investigates tech giants over social media ban breaches

-
 Hindu devotional clubbing sways India's youth
Hindu devotional clubbing sways India's youth
-
Oil slips, stocks rise as report says Trump willing to end war

-
 Mind games: How football stars are fuelling chess boom
Mind games: How football stars are fuelling chess boom
-
Indonesia trims meals programme: what next?

-
 'A very big deal': Canadian astronaut reflects on historic Moon mission
'A very big deal': Canadian astronaut reflects on historic Moon mission
-
US pro table tennis league blasts niche sport into spotlight

-
 Iran defiant as Trump threatens to destroy oil island
Iran defiant as Trump threatens to destroy oil island
-
Clint McKinlay Joins Good Driver Mutuality as President of Sales

-
 Wiley Launches "The Black Book of Reshoring: The Essential Guide to America's New Manufacturing Boom" by Douglas Brown
Wiley Launches "The Black Book of Reshoring: The Essential Guide to America's New Manufacturing Boom" by Douglas Brown
-
InterContinental Hotels Group PLC Announces Transaction in Own Shares - March 31

-
 Star Copper Advances Integrated 3D Geological Modeling to Position 2026 Drill Season As Transformational
Star Copper Advances Integrated 3D Geological Modeling to Position 2026 Drill Season As Transformational
-
Empire Metals Limited Announces Eclipse Mining Licence Sale Extension

-
 MindMaze Therapeutics and Vibra Healthcare Announce Breakthrough RWE Results in High-Dose, High-Intensity Neurorehabilitation
MindMaze Therapeutics and Vibra Healthcare Announce Breakthrough RWE Results in High-Dose, High-Intensity Neurorehabilitation
-
Trump threatens to destroy Iran oil island despite claims of talks

-
 NASA begins countdown to April 1 Moon launch
NASA begins countdown to April 1 Moon launch
-
NBA Bulls fire Ivey after anti-LGBTQ comments

-
 Australian regulator probes Facebook, YouTube over teen social media ban
Australian regulator probes Facebook, YouTube over teen social media ban
-
Iraq coach shielding players from war ahead of World Cup bid

-
 Undav rescues Germany late in Ghana friendly
Undav rescues Germany late in Ghana friendly
-
Messi to start for Argentina in World Cup send-off: Scaloni

-
 Oil rises on Trump's Iran threats, stocks mixed
Oil rises on Trump's Iran threats, stocks mixed
-
After pope's remark, White House defends praying for US troops

-
 Powell probe leaves US Fed leadership change in limbo
Powell probe leaves US Fed leadership change in limbo
-
Celine Dion announces comeback following health struggle

-
 'Is it Kafka?' US judge baffled by new Pentagon press policy
'Is it Kafka?' US judge baffled by new Pentagon press policy
-
Cubans ready for Russian oil but some say not enough

-
 Teen Suryavanshi shines as Rajasthan hammer Chennai in IPL
Teen Suryavanshi shines as Rajasthan hammer Chennai in IPL
-
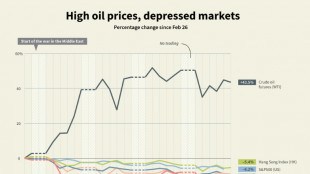
Stock market winners and losers one month into US-Israel war on Iran
-
 Hodgson says surprise return to management is only for short-term
Hodgson says surprise return to management is only for short-term
-
What could Trump achieve by threatening Iran's Kharg Island?

-
 India declares victory over Maoist insurgency
India declares victory over Maoist insurgency
-
Germany's Merz pushes return of Syrians as he hosts leader Sharaa


US approves new drug to treat Alzheimer's disease
The US Food and Drug Administration on Friday approved a highly anticipated new drug designed to slow cognitive decline in patients in mild and early stages of Alzheimer's disease.
The FDA approval of the drug, Leqembi, also known as lecanemab, comes just days after the regulatory agency was harshly criticized in a congressional report for its green-lighting of another Alzheimer's drug, Aduhelm.
And it was granted despite trial results showing the monoclonal antibody treatment carries risks of brain swelling and bleeding.
Both drugs were approved through an accelerated process that allows the FDA to fast-track approval of drugs for serious conditions where there is an unmet medical need.
Leqembi and Aduhelm, which were jointly developed by Japan's Eisai and Biogen of the United States, "represent an important advancement in the ongoing fight to effectively treat Alzheimer's disease," the FDA said in a statement.
"Alzheimer's disease immeasurably incapacitates the lives of those who suffer from it and has devastating effects on their loved ones," Billy Dunn of the FDA's Center for Drug Evaluation and Research said in a statement.
Leqembi, Dunn said, is "the latest therapy to target and affect the underlying disease process of Alzheimer's, instead of only treating the symptoms of the disease."
Approximately 6.5 million Americans suffer from Alzheimer's, which is characterized by memory loss and declining mental acuity.
Preliminary data from a trial of Leqembi was released in September and found it slowed cognitive decline in Alzheimer's patients by 27 percent.
The phase three trial involved nearly 1,800 people, divided between those given the drug and given a placebo, and ran over 18 months.
The complete trial data, published in the New England Journal of Medicine, raised concern about the incidence of "adverse effects" including brain bleeds and swelling.
The results showed that 17.3 percent of patients administered the drug experienced brain bleeds, compared with nine percent of those receiving a placebo.
And 12.6 percent of those taking the drug experienced brain swelling, compared with just 1.7 percent of those in the placebo group.
Deaths were reported at approximately the same rate in both arms of the trial of the drug.
- $26,500 a year -
In Alzheimer's disease, two key proteins, tau and amyloid beta, build up into tangles and plaques, known together as aggregates, which cause brain cells to die and lead to brain shrinkage.
Leqembi, which is administered intravenously once every two weeks, works by targeting amyloid.
In the trial, patients receiving Leqembi had a statistically significant reduction in brain amyloid plaque compared to the placebo arm, which had no reduction of amyloid beta plaque.
Biogen and Eisai previously brought Aduhelm to market, but there was significant controversy over whether it worked, and its approval in 2021 led to three high-level resignations in the FDA.
A US congressional investigation said the accelerated approval process for Aduhelm, the first drug approved in decades to treat Alzheimer's, was "rife with irregularities" and criticized both the agency and Biogen.
The Cambridge, Massachusetts-based Biogen set an "unjustifiably high price" for Aduhelm of $56,000 a year, the congressional report said.
Eisai said Leqembi would be priced initially at $26,500 per year and estimated that 100,000 Americans could be receiving the drug by three years from now.
Joanne Pike, president and CEO of the Alzheimer's Association, welcomed the approval of Leqembi but expressed concern that its high cost could put it out of reach of most Americans, particularly if it is not covered by Medicare, the government health insurance program for the elderly.
"People living with this fatal disease today do not have time to wait for a miracle drug or cure," Pike said in a statement.
Y.Baker--AT




