
-
 Second unexploded shell found at illegal French rave: minister
Second unexploded shell found at illegal French rave: minister
-
Bournemouth eye European place after crushing Palace

-
 Pogacar ends dominant Tour of Romandie with fourth win
Pogacar ends dominant Tour of Romandie with fourth win
-
Chakravarthy, Narine help Kolkata stay alive in IPL

-
 Daughter says Maradona died after carers' plan 'went out of control'
Daughter says Maradona died after carers' plan 'went out of control'
-
Two women suffocate on migrant boat seeking to reach UK

-
 How Schalke returned to the Bundesliga after their 'worst season ever'
How Schalke returned to the Bundesliga after their 'worst season ever'
-
Two women die on migrant boat seeking to reach UK

-
 Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'
Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'
-
Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach

-
 Bangkok food vendor curbs push city staple from the streets
Bangkok food vendor curbs push city staple from the streets
-
More Nepalis drive electric, evading global fuel shocks

-
 Latecomer Japan eyes slice of rising global defence spending
Latecomer Japan eyes slice of rising global defence spending
-
Messi goal not enough as Miami collapse in 4-3 loss to Orlando

-
 German fertiliser makers and farmers struggle with Iran war fallout
German fertiliser makers and farmers struggle with Iran war fallout
-
OPEC+ to make first post-UAE production decision

-
 Massive crowds fill Rio's Copacabana beach for Shakira concert
Massive crowds fill Rio's Copacabana beach for Shakira concert
-
Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs

-
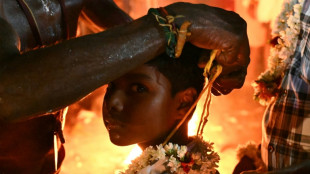 Fleeting freedom at festival for India's transgender community
Fleeting freedom at festival for India's transgender community
-
Trump says cutting US troop numbers in Germany 'way down'

-
 Man charged with murdering Indigenous girl in Australian outback
Man charged with murdering Indigenous girl in Australian outback
-
CMS EXPOSED: The "Workaround Doctrine" - How Matt Zorn's Legal CMS Hemp Strategy Undermines the FDA

-
 Prometheus Signals Near-Term Development of Next-Generation Noninvasive Test for MASH Patients at DDW 2026
Prometheus Signals Near-Term Development of Next-Generation Noninvasive Test for MASH Patients at DDW 2026
-
China's Wu Yize wins last-frame thriller to reach snooker world final

-
 Serene Korda takes three-shot lead at LPGA Mexico
Serene Korda takes three-shot lead at LPGA Mexico
-
Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux

-
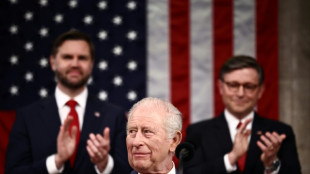 King Charles grasped 'opportunity' on US trip, palace says
King Charles grasped 'opportunity' on US trip, palace says
-
China's Wu wins last-frame thriller to reach snooker world final

-
 Verstappen sees light at the end of tunnel
Verstappen sees light at the end of tunnel
-
Young stretches PGA lead to six at Doral

-
 Rio's Copacabana beach hosts massive crowd for free Shakira concert
Rio's Copacabana beach hosts massive crowd for free Shakira concert
-
Celtics' Tatum ruled out for decisive game seven against Sixers

-
 Wolff heralds Antonelli speed as teen joins Senna and Schumacher in record books
Wolff heralds Antonelli speed as teen joins Senna and Schumacher in record books
-
Senior Iranian officer says fresh conflict with US 'likely'

-
 Barcelona on verge of Liga title, Villarreal secure top four
Barcelona on verge of Liga title, Villarreal secure top four
-
Teen F1 leader Antonelli takes Miami Grand Prix pole

-
 Porto edge Alverca to clinch Portuguese league title
Porto edge Alverca to clinch Portuguese league title
-
US airlines step up as Spirit winds down

-
 Barcelona on verge of La Liga title defence with win at Osasuna
Barcelona on verge of La Liga title defence with win at Osasuna
-
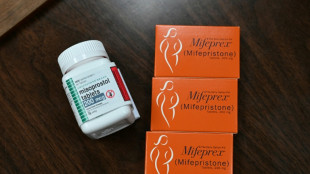
Drugmaker asks US Supreme Court to restore abortion pill access
-
 Schalke return to Bundesliga after three-year absence
Schalke return to Bundesliga after three-year absence
-
NATO, top Republicans question US troop withdrawal from Germany

-
 Napoli frustrate Como in costly Serie A stalemate
Napoli frustrate Como in costly Serie A stalemate
-
Illegal party at French military site draws up to 40,000 ravers

-
 Arsenal hit stride to go six points clear, West Ham loss offers Spurs hope
Arsenal hit stride to go six points clear, West Ham loss offers Spurs hope
-
Arsenal go six points clear as Gyokeres double sinks Fulham

-
 Clinical Chennai down Mumbai to keep playoff hopes alive
Clinical Chennai down Mumbai to keep playoff hopes alive
-
Napoli and Como play out goalless draw in Serie A

-
 Murphy into World Snooker Championship final after edging Higgins
Murphy into World Snooker Championship final after edging Higgins
-
PSG held by Lorient with fringe team ahead of Bayern Munich return leg


CMS EXPOSED: The "Workaround Doctrine" - How Matt Zorn's Legal CMS Hemp Strategy Undermines the FDA
Matthew C. Zorn previously argued that federal regulators determine "accepted medical use" through the FDA approval framework. Today, he appears on behalf of the United States defending a CMS program that introduces cannabinoid furnishing into Medicare connected clinical environments without FDA validation. The contradiction raises serious policy questions: if FDA approval defines medical legitimacy under federal law, how can Medicare linked access pathways expand before that standard is met? Programs like BEI risk signaling that clinical evidence is optional - a message that directly undermines companies investing years in FDA authorized cannabinoid drug development.
WASHINGTON, D.C. / ACCESS Newswire / May 3, 2026 / A major federal lawsuit now pending in the U.S. District Court for the District of Columbia is raising urgent questions about whether cannabinoid products can enter Medicare connected healthcare environments before they are proven safe and effective through the federal drug approval process.

At the center of the dispute is:
Smart Approaches to Marijuana (SAM), et al. v. Robert F. Kennedy Jr., et al.
Case No. 1:26-cv-01081 (D.D.C.)
The case challenges the legality of the Centers for Medicare & Medicaid Services' Beneficiary Engagement Incentive (BEI) model - an Innovation Center program allowing provider-mediated furnishing of certain hemp-derived cannabinoid products to Medicare beneficiaries without approval from the U.S. Food and Drug Administration.
Critics say the policy represents something unprecedented:
Medicare infrastructure moving ahead of medicine.
A Structural Break From the Federal Drug Approval Sequence
For decades, federal healthcare policy followed a consistent rule:
science → clinical trials → FDA approval → coverage integration
The BEI model reverses that order:
distribution → furnishing → infrastructure exposure → evidence later
That shift is not procedural housekeeping.
It is a policy pivot.
The Lawsuit That Could Define the Boundary Between CMS and FDA Authority
The plaintiffs in SAM et al. v. Kennedy argue the Centers for Medicare & Medicaid Services exceeded its authority by introducing cannabinoid furnishing pathways into federally funded care environments without traditional safeguards such as:
• Federal Register rulemaking
• notice-and-comment procedures
• FDA drug approval
• interstate therapeutic compliance standards
Historically, the role of the Centers for Medicare & Medicaid Services has been to administer coverage - not determine what qualifies as medicine.
That responsibility belongs to the FDA.
Matthew C. Zorn Now Appears on Behalf of the United States
Federal filings identify:
Matthew C. Zorn
Deputy General Counsel
U.S. Department of Health and Human Services
Special Assistant U.S. Attorney
as appearing in the litigation for the United States government.
The development is drawing attention because Zorn previously argued publicly that federal regulators determine "accepted medical use" through the FDA approval framework.
Now he appears defending a policy structure critics say allows cannabinoid products to enter Medicare-connected clinical environments without satisfying that same standard.
Observers across the pharmaceutical cannabinoid sector are asking:
Has federal policy quietly shifted from evidence-first medicine to infrastructure-first access?
Why Clinical-Stage Cannabinoid Developers Are Alarmed
Companies pursuing cannabinoid therapies through FDA pathways invest years completing:
• Investigational New Drug authorizations
• chemistry, manufacturing, and controls validation
• stability testing programs
• toxicology and safety datasets
• controlled-substance compliance obligations
• orphan-designation development strategies
Programs like BEI risk creating a powerful new signal:
clinical trials are optional
That message could reshape the economics of cannabinoid drug development nationwide.
The Quiet Creation of a Parallel Therapeutic Channel
The CMS Innovation Center has broad authority to test payment and delivery models.
It was never designed to function as a substitute therapeutic approval pathway.
Yet the BEI structure introduces cannabinoid furnishing into Medicare-connected care environments without FDA validation.
Policy analysts warn this risks creating:
a shadow cannabinoid healthcare system operating outside the evidence-based drug framework
Why the Timing Makes the Case Especially Important
The lawsuit arrives at a turning point in federal cannabinoid regulation:
• cannabis scheduling reform remains under active federal review
• DEA research-licensing bottlenecks are under scrutiny
• botanical drug-development pathways are expanding
• orphan-designation cannabinoid therapies remain active
• Medicare cannabinoid coverage policy remains unsettled
Introducing reimbursement-adjacent cannabinoid furnishing into this environment without FDA approval risks destabilizing the clinical-evidence pipeline supporting pharmaceutical cannabinoid innovation.
The Incentive Problem No One in Washington Is Addressing
Federal drug development depends on a predictable rule:
evidence comes before reimbursement
If reimbursement infrastructure begins appearing first, the incentive to conduct expensive clinical trials weakens dramatically.
That affects:
patient safety
manufacturing consistency
dose reliability
interstate labeling integrity
long-term pharmacovigilance systems
In short:
it changes how medicine enters the healthcare system.
The Legal Question Now Before the Court
The outcome of:
Smart Approaches to Marijuana (SAM), et al. v. Robert F. Kennedy Jr., et al.
Case No. 1:26-cv-01081 (D.D.C.)
may determine whether the CMS Innovation Center can function as a parallel entry pathway for cannabinoid therapeutics outside the FDA approval structure.
That decision will shape more than cannabinoid policy.
It will define whether Medicare infrastructure can move ahead of medicine itself.
The Central Question Facing Federal Regulators
Matthew C. Zorn once helped challenge federal barriers limiting cannabis research.
Today, as Deputy General Counsel at HHS and Special Assistant U.S. Attorney, he appears on behalf of the United States defending federal authority to implement a cannabinoid furnishing model critics say bypasses the clinical-evidence pathway.
The question now confronting policymakers is straightforward:
Will cannabinoid therapies enter Medicare because they have been proven safe and effective through science-
or because someone found a way around the rules?
Madison Hisey
[email protected]
203-231-85832
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
A.Clark--AT
