
-
 Rights groups fear use of arrest to stifle free speech in Pakistan
Rights groups fear use of arrest to stifle free speech in Pakistan
-
Iranian missiles sow panic, destruction in Israeli towns

-
 Damaged Russian tanker to be towed to Libya: state-owned company
Damaged Russian tanker to be towed to Libya: state-owned company
-
Gilgeous-Alexander scores 40, LeBron breaks NBA appearance record

-
 Cuba hit by second nationwide blackout in a week
Cuba hit by second nationwide blackout in a week
-
BTS draws over 100,000 fans to Seoul comeback concert: label

-
 US-China 'Board of Trade' may help ties but experts flag market worries
US-China 'Board of Trade' may help ties but experts flag market worries
-
Sinner, defending champ Mensik advance to third round at Miami Open

-
 Iran missile strikes wound over 100 in two south Israel towns
Iran missile strikes wound over 100 in two south Israel towns
-
Shai hits 40 as Thunder win despite NBA melee with four ejected

-
 Records shattered as US heatwave moves eastward
Records shattered as US heatwave moves eastward
-
Iran missiles hit southern Israel, injuring more than 100

-
 LeBron James breaks record for most NBA games played
LeBron James breaks record for most NBA games played
-
'Perfect' PSG sweep past Nice to reclaim top spot in Ligue 1

-
 Japan coach says Asian Cup crown 'well-deserved' for inspirational team
Japan coach says Asian Cup crown 'well-deserved' for inspirational team
-
PSG sweep past Nice to reclaim top spot in Ligue 1

-
 Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81
Robert Mueller, ex-FBI chief who led Trump-Russia probe, dead at 81
-
Milan move to within five points of Serie A leaders Inter

-
 Duplantis masterclass as Kerr and record-setter Ehammer shine
Duplantis masterclass as Kerr and record-setter Ehammer shine
-
Rosenior urges Chelsea to 'forget the noise' after damaging loss

-
 Marquez ambushed Di Giannantonio to win Brazil sprint
Marquez ambushed Di Giannantonio to win Brazil sprint
-
Sweden's Duplantis wins fourth world indoor pole vault title

-
 Liverpool, Chelsea slip up in Champions League race
Liverpool, Chelsea slip up in Champions League race
-
WHO sends first overland convoy from emergencies hub to Beirut

-
 Everton rub salt in Chelsea wounds as Champions League race tightens
Everton rub salt in Chelsea wounds as Champions League race tightens
-
Coach Mignoni returns but Toulon crash to Stade Francais

-
 Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81
Robert Mueller, ex-FBI chief who led Trump-Russia inquiry, dead at 81
-
Sinner and Pegula advance to third round at Miami Open

-
 Britain's Kerr outsprints Hocker for world indoor 3,000m gold
Britain's Kerr outsprints Hocker for world indoor 3,000m gold
-
Kane backs Tuchel's call to rest him from England friendly

-
 NBA fines 76ers' Drummond, Magic's Suggs $25,000 each
NBA fines 76ers' Drummond, Magic's Suggs $25,000 each
-
Switzerland's Ehammer sets indoor heptathlon world record

-
 Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set
Pogacar 'relieved' by Milan-San Remo triumph, gunning to complete Monument set
-
World Athletics decision to hand Asia two world indoors 'strategic' - Coe

-
 Trump threatens to use ICE agents for airport security control
Trump threatens to use ICE agents for airport security control
-
Kane moves closer to goals record as Bayern sink Union

-
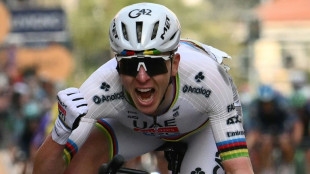 Pogacar ends long wait for Milan-San Remo glory after edging epic
Pogacar ends long wait for Milan-San Remo glory after edging epic
-
US says 'took out' Iran base threatening blocked Hormuz oil route

-
 Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez
Di Giannantonio takes Brazil MotoGP pole ahead of Bezzecchi, Marquez
-
Welbeck scores twice to dent Liverpool's top-five hopes

-
 US strikes Iran bases threatening blocked Hormuz oil route
US strikes Iran bases threatening blocked Hormuz oil route
-
Pirovano wins World Cup downhill title, Aicher puts pressure on Shiffrin

-
 Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast
Doroshchuk wins Ukraine's second world indoor gold, Hodgkinson and Alfred coast
-
K-pop kings BTS stun Seoul in '2.0' comeback concert

-
 French prosecutors suspect Musk encouraged deepfakes row to inflate X value
French prosecutors suspect Musk encouraged deepfakes row to inflate X value
-
Mbappe 100 percent, Bellingham fit, says Real Madrid's Arbeloa

-
 Iranians mark Eid as Tehran reports strike on nuclear plant
Iranians mark Eid as Tehran reports strike on nuclear plant
-
Kenya, Uganda open rail extension burdened by Chinese debt

-
 K-pop kings BTS rock Seoul in comeback concert
K-pop kings BTS rock Seoul in comeback concert
-
Invincible Japan edge Australia to win Women's Asian Cup


The Coming Divide in Cannabis: Why the U.S. Market Remains Out of Reach for Global Operators
"For too long, legitimate cannabinoid medicine has been trapped between two federal systems that were never designed to work together," said Duane Boise, CEO of MMJ International Holdings. "The FDA encourages clinical research into cannabinoids, yet companies pursuing those trials have faced extraordinary obstacles obtaining the pharmaceutical-grade materials required to conduct them. If the United States is serious about developing cannabinoid medicines, the regulatory framework must support science rather than obstruct it."
WASHINGTON, DC / ACCESS Newswire / March 5, 2026 / Most headlines about cannabis policy focus on legalization, tax revenue, or retail sales. But beneath the surface, a far more consequential battle has been unfolding inside the federal regulatory system-one that could ultimately determine who controls the future of cannabinoid medicine in the United States.

While thousands of companies built businesses around state cannabis markets, MMJ pursued a far more difficult path: developing FDA-regulated cannabinoid medicines through formal clinical trials.
That decision placed the company directly inside a complex regulatory intersection involving the U.S. Food and Drug Administration, the Drug Enforcement Administration, and the federal courts.
And it is that intersection that could reshape the entire industry.
The Core Issue: Who Controls Medical Cannabis Research?
Under federal law, any company seeking to develop cannabinoid-based medicines must navigate two separate regulatory systems:
FDA drug development oversight
DEA control over controlled substances and manufacturing registrations
This dual-agency structure has historically created significant bottlenecks for legitimate medical research.
For years, the DEA maintained strict limits on who could cultivate cannabis for research, leaving many FDA-authorized clinical programs without adequate supply of pharmaceutical-grade material.
The result was a regulatory paradox:
The FDA encouraged scientific research into cannabinoids
But researchers often could not obtain the material needed to conduct those studies.
Why the MMJ Case Matters
The dispute surrounding MMJ's DEA registration application highlights this contradiction more clearly than any previous case.
Unlike most cannabis companies, MMJ pursued the formal pharmaceutical pathway, including FDA-authorized clinical development programs targeting serious neurological diseases such as:
Huntington's disease
Multiple sclerosis
These programs rely on consistent, pharmaceutical-grade cannabinoid formulations - not the variable plant material commonly sold in retail dispensaries.
The company's legal challenge argues that federal regulatory delays have effectively prevented the development of legitimate cannabinoid medicines, despite congressional directives intended to expand research.
Why Global Cannabis Companies Still Cannot Enter the U.S. Market
While much of the North American cannabis industry has focused on retail expansion, some of the largest Canadian licensed producers have begun pivoting toward regulated medical markets abroad.
Across Europe, countries such as Germany and Poland have developed structured national medical cannabis systems that operate more like pharmaceutical distribution networks than retail markets.
These systems emphasize:
physician-driven prescriptions
regulated supply chains
standardized medical products
But despite their scale and international reach, even the largest Canadian cannabis producers face a fundamental barrier when it comes to the United States.
The reason is simple: the federal pharmaceutical pathway remains the only fully lawful route into the U.S. cannabinoid medicine market.
Without:
FDA drug approval
federally authorized manufacturing
DEA research registration
foreign cannabis companies cannot legally participate in the American medical cannabinoid sector at scale.
As a result, global operators seeking to access the U.S. market must either partner with companies already inside the federal regulatory framework or pursue their own multi-year FDA drug development programs.
A Potential Turning Point for Federal Cannabis Policy
If the courts ultimately determine that the DEA improperly blocked or delayed legitimate pharmaceutical research registrations, the consequences could be profound.
Such a ruling could force structural changes in how cannabis research is regulated, including:
expanding the number of federally authorized cannabis manufacturers
accelerating supply access for clinical trials
aligning DEA policy with FDA-approved research programs
In effect, the case could transform the federal cannabis research system from a limited experimental program into a functioning pharmaceutical supply chain.
Why This Matters for the Entire Industry
For most cannabis companies, the stakes extend far beyond a single registration decision.
If federal policy fully embraces the pharmaceutical model for cannabinoid therapies, the industry could undergo a dramatic realignment.
Future cannabinoid medicines will require:
FDA clinical trials
standardized pharmaceutical formulations
regulated manufacturing and distribution
This would shift the center of gravity of the cannabis sector away from retail dispensaries and toward biotechnology companies capable of navigating federal drug development pathways.
The precedent was already established when the FDA approved Epidiolex, proving that cannabinoid medicines can reach full regulatory approval.
But the next phase of the industry will depend on whether the federal system allows additional companies to follow that same path.
A Defining Moment for Cannabinoid Medicine
The outcome of the MMJ case may ultimately determine whether the United States develops a robust pharmaceutical cannabinoid sector or continues to rely on fragmented state markets.
If the courts force regulatory alignment between federal agencies and FDA-authorized research programs, the ripple effects could reshape the industry for decades.
In that scenario, the legal battle surrounding MMJ would not simply be remembered as a dispute over a single license.
It would be seen as the moment when the American pharmaceutical cannabis industry truly began.
CONTACT:
Madison Hisey
[email protected]
203-231-8583
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
N.Walker--AT
