
-
 Oil spill snarls shipping traffic in Antwerp port
Oil spill snarls shipping traffic in Antwerp port
-
Giving birth in a shelter in Israel
-
 Five things to know about the planned Iran-US talks in Islamabad
Five things to know about the planned Iran-US talks in Islamabad
-
Slot feels 'complete support' from Liverpool chiefs despite slump

-
 Kyiv books tentative diplomatic coup with Iran war forays
Kyiv books tentative diplomatic coup with Iran war forays
-
Teenager shines as Britain seize control of BJK Cup tie with Australia

-
 Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader
Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader
-
Sleepy seal diverts traffic in Australian seaside town

-
 Artemis astronauts to shed light on space health risks
Artemis astronauts to shed light on space health risks
-
Pakistan prepares to host US-Iran talks, as Lebanon fighting continues

-
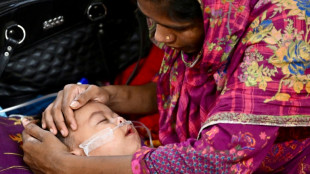 Vaccine gaps fuel Bangladesh's deadly measles crisis
Vaccine gaps fuel Bangladesh's deadly measles crisis
-
Fish furore fuels fierce election in India's West Bengal

-
 Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G
Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G
-
Myanmar junta chief sworn in as president

-
 Exiled cartoonists give voice to Iran's silenced millions
Exiled cartoonists give voice to Iran's silenced millions
-
In Pakistan's mediation to end Mideast war, China may hold the key

-
 Knicks stay in hunt with late win over rival Celtics
Knicks stay in hunt with late win over rival Celtics
-
'Sartorial diplomacy' on show in expo of late UK queen's fashion

-
 Former Japan and AC Milan star Honda laces up boots again at 39
Former Japan and AC Milan star Honda laces up boots again at 39
-
Stocks rally on optimism over Iran war ceasefire, oil extends gains

-
 Lego-style memes troll Trump after fragile US-Iran truce
Lego-style memes troll Trump after fragile US-Iran truce
-
Chinese slimmers trade lost fat for beef
-
 Jackson biopic shows franchise thriving despite abuse claims
Jackson biopic shows franchise thriving despite abuse claims
-
New Jersey city spurns data center as defiance spreads

-
 US box office looking good as cinema owners gather: industry chief
US box office looking good as cinema owners gather: industry chief
-
Firm Masters greens make life hard on golf's finest

-
 Defending champ McIlroy shares Masters lead after back-nine birdie run
Defending champ McIlroy shares Masters lead after back-nine birdie run
-
After oil, Venezuela opens up mining to private investors
-
 Tigers' Meadows in hospital after colliding with teammate
Tigers' Meadows in hospital after colliding with teammate
-
US to host Israel-Lebanon talks as strikes threaten Iran ceasefire

-
 'Scrappy' McIlroy leans on experience for share of Masters lead
'Scrappy' McIlroy leans on experience for share of Masters lead
-
Ukraine and Russia will cease fire for Orthodox Easter

-
 Mateta inspires Palace win over Fiorentina in Conference League
Mateta inspires Palace win over Fiorentina in Conference League
-
Pioneering US hip-hop artist Afrika Bambaataa dies at 68

-
 Russia bans Nobel-winning rights group, raids independent newspaper, in one day
Russia bans Nobel-winning rights group, raids independent newspaper, in one day
-
Pentagon denies giving Vatican envoy 'bitter lecture'

-
 Watkins propels Villa towards Europa League semis, Forest hold Porto
Watkins propels Villa towards Europa League semis, Forest hold Porto
-
Aston Villa on verge of Europa League semis after beating Bologna

-
 Venezuela police clash with protesters demanding salary rises
Venezuela police clash with protesters demanding salary rises
-
CAF president rejects corruption claims by Senegal

-
 Israel and Lebanon set for ceasefire talks next week, says US official
Israel and Lebanon set for ceasefire talks next week, says US official
-
US stocks extend gains, shrugging off ceasefire worries

-
 IMF chief urges nations to 'do no harm' in fiscal response to Iran war
IMF chief urges nations to 'do no harm' in fiscal response to Iran war
-
Sixers' Embiid to have surgery for appendicitis - team

-
 Russian police raid independent Novaya Gazeta outlet, reporter detained
Russian police raid independent Novaya Gazeta outlet, reporter detained
-
Former heavyweight king Fury adamant 'I've still got it' as Makhmudov awaits

-
 Shipping toll for Hormuz passage sharply divides nations
Shipping toll for Hormuz passage sharply divides nations
-
McIlroy's back-nine birdie run grabs share of Masters lead

-
 Melania Trump blasts 'lies' linking her to Epstein
Melania Trump blasts 'lies' linking her to Epstein
-
'Anxious' Tatum back at Madison Square Garden with NBA East second seed on line


Jaguar Health Reports Approval of All Proposals at December 2025 Special Meeting of Stockholders
Jaguar CEO Lisa Conte presenting December 10 from 3:25pm - 3:35pm Eastern at the Emerging Growth Conference to provide updates on near-term catalysts; Click here to register
Initial results of ongoing investigator-initiated proof-of-concept trial demonstrate crofelemer can potentially extend the lives of infants born with intestinal failure due to microvillus inclusion disease (MVID), reducing the volume of the total parenteral support (PS) necessary for them to survive, though with associated toxicity; Groundbreaking PS reduction of up to 37%; No approved treatments exist for MVID
Company completed meeting with FDA October 2, 2025 for advice on advancing its ongoing placebo-controlled trial of crofelemer in support of possible expedited approval pathway for crofelemer for treatment of MVID
SAN FRANCISCO, CALIFORNIA / ACCESS Newswire / December 8, 2025 / Jaguar Health, Inc. (NASDAQ:JAGX) ("Jaguar" or the "Company") today announced the voting results of the Company's Special Meeting of Stockholders held on December 8, 2025 (the "Special Meeting").
Three proposals were submitted to and approved by the stockholders of the Company at the Special Meeting. The proposals are described in detail in the Company's definitive proxy statement on Schedule 14A relating to the Special Meeting filed with the Securities and Exchange Commission on November 10, 2025. Stockholders may obtain a free copy of the proxy statement and other documents filed by Jaguar with the SEC at http://www.sec.gov. The proxy statement is also available on the Company's corporate website.
About the Jaguar Health Family of Companies
Jaguar Health, Inc. (Jaguar) is a commercial stage pharmaceuticals company focused on developing novel proprietary prescription medicines sustainably derived from plants from rainforest areas for people and animals with gastrointestinal distress, specifically associated with overactive bowel, which includes symptoms such as chronic debilitating diarrhea, urgency, bowel incontinence, and cramping pain. Jaguar family company Napo Pharmaceuticals (Napo) focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states. Napo's crofelemer is FDA-approved under the brand name Mytesi® for the symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy. Jaguar family company Napo Therapeutics is an Italian corporation Jaguar established in Milan, Italy in 2021 focused on expanding crofelemer access in Europe and specifically for orphan diseases. Jaguar Animal Health is a Jaguar tradename. Magdalena Biosciences, a joint venture formed by Jaguar and Filament Health Corp. that emerged from Jaguar's Entheogen Therapeutics Initiative (ETI), is focused on developing novel prescription medicines derived from plants for mental health indications.
For more information about:
Jaguar Health, visit https://jaguar.health
Napo Pharmaceuticals, visit www.napopharma.com
Napo Therapeutics, visit napotherapeutics.com
Magdalena Biosciences, visit magdalenabiosciences.com
Canalevia-CA1, visit canalevia.com
Visit the Make Cancer Less Shitty patient advocacy program on Bluesky, X, Facebook & Instagram
Forward-Looking Statements
Certain statements in this press release constitute "forward-looking statements." These include statements regarding Jaguar's expectation that Jaguar management will present at the December 2025 Emerging Growth Conference, Jaguar's expectation that crofelemer can potentially extend the lives of infants born with intestinal failure due to MVID, reducing the volume of PS necessary for them to survive, and Jaguar's expectation that an expedited approval pathway may be possible for crofelemer for treatment of MVID. In some cases, you can identify forward-looking statements by terms such as "may," "will," "should," "expect," "plan," "aim," "anticipate," "could," "intend," "target," "project," "contemplate," "believe," "estimate," "predict," "potential" or "continue" or the negative of these terms or other similar expressions. The forward-looking statements in this release are only predictions. Jaguar has based these forward-looking statements largely on its current expectations and projections about future events. These forward-looking statements speak only as of the date of this release and are subject to several risks, uncertainties, and assumptions, some of which cannot be predicted or quantified and some of which are beyond Jaguar's control. Except as required by applicable law, Jaguar does not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
Source: Jaguar Health, Inc.
Contact:
Jaguar-JAGX
SOURCE: Jaguar Health, Inc.
View the original press release on ACCESS Newswire
J.Gomez--AT
