
-
 Trump makes stark threat to Iran after US airman rescued
Trump makes stark threat to Iran after US airman rescued
-
Artemis astronauts ready for Moon flyby on fifth day of historic mission

-
 Israel renews Lebanon strikes, forces Syria border crossing closed
Israel renews Lebanon strikes, forces Syria border crossing closed
-
Eagle-eyed Spaun snatches Texas Open victory

-
 Brown, Tatum propel Celtics in win over Raptors
Brown, Tatum propel Celtics in win over Raptors
-
Paul battles past Burruchaga to win ATP Houston title

-
 Major sponsors drop Kanye West London gigs as PM voices concern
Major sponsors drop Kanye West London gigs as PM voices concern
-
Inter close in on Serie A title by thumping Roma

-
 Trump makes foul-mouthed threat to Iran after US airman rescued
Trump makes foul-mouthed threat to Iran after US airman rescued
-
Monaco sink Marseille for seventh Ligue 1 win in a row

-
 Inter thump Roma to extend Serie A lead to nine points
Inter thump Roma to extend Serie A lead to nine points
-
Lebanon's Christians mark Easter in solidarity with war-hit south

-
 Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years
Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years
-
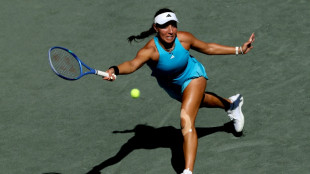
Pegula romps to WTA Charleston Open victory
-
 David six-hitting spree powers Bengaluru to IPL win
David six-hitting spree powers Bengaluru to IPL win
-
Union draw leaves St Pauli stranded in Bundesliga drop zone

-
 UK police arrest protesters near base used by US
UK police arrest protesters near base used by US
-
Trump issues foul-mouthed threat to Iran after US airman rescued

-
 Alcaraz plans to play full clay-court season, get 'socks dirty'
Alcaraz plans to play full clay-court season, get 'socks dirty'
-
'Super Mario Galaxy' blasts off in N. America box office debut

-
 Artemis astronauts begin fifth day on historic Moon mission
Artemis astronauts begin fifth day on historic Moon mission
-
Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup

-
 Trump draws criticism with fiery Easter message on Iran
Trump draws criticism with fiery Easter message on Iran
-
OPEC+ hikes oil production quotas, issues warning

-
 British PM slams London event for booking Kanye West, sponsor quits
British PM slams London event for booking Kanye West, sponsor quits
-
Pogacar wins joint-record third Tour of Flanders

-
 Trump threatens 'hell' for Iran over Strait of Hormuz
Trump threatens 'hell' for Iran over Strait of Hormuz
-
Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash

-
 What we know about the race to rescue downed US airman in Iran
What we know about the race to rescue downed US airman in Iran
-
US commandos went deep into Iran to rescue downed airman: media

-
 Liberated McIlroy eyes more Masters magic after career Slam
Liberated McIlroy eyes more Masters magic after career Slam
-
Van Dijk apologises for Liverpool thumping by Man City

-
 British PM slams London festival for booking Kanye West
British PM slams London festival for booking Kanye West
-
'Choose peace': Pope marks first Easter under cloud of Mideast war

-
 British royals attend Easter service without Andrew
British royals attend Easter service without Andrew
-
US media says commandos probed deep into Iran to rescue downed airman

-
 Revellers parade giant penises to dash stigma in Japan's fertility festival
Revellers parade giant penises to dash stigma in Japan's fertility festival
-
Artemis astronauts glimpse Moon's 'Grand Canyon' ahead of historic lunar flyby

-
 Middle East war hits Britain's fish and chip shops
Middle East war hits Britain's fish and chip shops
-
Artemis astronauts to study the Moon's surface using mainly their eyes

-
 Second US airman downed over Iran 'SAFE and SOUND': Trump
Second US airman downed over Iran 'SAFE and SOUND': Trump
-
Indonesia lays to rest peacekeepers killed in Lebanon

-
 Pharmaceutical logistics in demand as war rattles supply chains
Pharmaceutical logistics in demand as war rattles supply chains
-
Messi marks new stadium with goal but Miami held by Austin

-
 Afghan mother seeks justice after Pakistani bombing kills hundreds
Afghan mother seeks justice after Pakistani bombing kills hundreds
-
UK royal family's dilemma over Andrew's daughters

-
 Pope marks first Easter under cloud of Mideast war
Pope marks first Easter under cloud of Mideast war
-
AI at war: Five things to know about Project Maven

-
 In the online 'maxxing' era, what's the deal with fiber and protein?
In the online 'maxxing' era, what's the deal with fiber and protein?
-
At Met Opera, life after a school shooting takes center stage


Ainos Reports Statistically Significant Interim Efficacy Results from VELDONA Clinical Trial in Feline Chronic Gingivostomatitis (FCGS)
Open-Label Study Demonstrates Durable Reduction in Oral Inflammation with Favorable Tolerability Profile at Interim Analysis
HOUSTON, TEXAS / ACCESS Newswire / February 17, 2026 / Ainos, Inc. (NASDAQ:AIMD)(NASDAQ:AIMDW) ("Ainos" or the "Company"), a SmellTech platform company digitizing scent as a native data language for artificial intelligence, today announced interim results from its ongoing open-label, multi-dose clinical trial evaluating VELDONA, a low-dose oral interferon alpha formulation, for the treatment of Feline Chronic Gingivostomatitis (FCGS), a severe, chronic inflammatory oral disease in companion animals.
The study is designed to enroll 12 client-owned cats diagnosed with FCGS across two dosing cohorts (6,000 IU and 12,000 IU administered orally three times per week for six weeks). The interim analysis includes the first six treated subjects. Enrollment remains ongoing.
Interim Efficacy Results
The primary disease activity assessment utilized the Stomatitis Disease Activity Index (SDAI; range 0-30), a composite score integrating:
Veterinary assessment of oral inflammation
Body weight changes
Owner-reported activity and feeding behavior
At Week 6:
The majority of subjects demonstrated reductions in total SDAI scores.
Directional improvement in overall disease activity was observed across the cohort.
No clinically significant rebound was observed through Week 14 follow-up.
Local oral inflammation scores demonstrated consistent and statistically significant improvement:
6 of 6 cats showed lower oral inflammation scores at Week 6 compared to baseline.
Mean improvement reached approximately 38% at Week 6.
66.7% of subjects achieved ≥30% clinically meaningful improvement.
Wilcoxon signed-rank testing demonstrated statistical significance (p
At Week 14 follow-up, mean improvement in oral inflammation increased to approximately 45%, suggesting sustained benefit after completion of the six-week treatment course.
Notably, one subject that did not undergo full-mouth dental extraction achieved measurable clinical improvement during interferon therapy, supporting a therapeutic effect independent of surgical recovery and consistent with an immunomodulatory mechanism.
Ainos plans to continue enrollment toward the full 12-subject target and further evaluate dose-response relationships, durability of effect, and safety outcomes in the complete dataset.
Safety and Tolerability
Comparative hematology and serum biochemistry analyses between baseline (Week 0) and Week 6 demonstrated:
Liver enzymes (ALP, ALT, AST) remained within normal limits.
Renal function markers (BUN, creatinine) remained stable without dose-related deterioration.
No persistent leukocyte abnormalities or evidence of immunosuppression were observed.
No dose-related toxicity trends were observed.
VELDONA™ was well tolerated in all six subjects, with no clinically significant safety signals identified in this interim analysis.
Study Overview
Design: Open-label, multi-dose clinical study
Population: Client-owned cats diagnosed with FCGS
Dosing: 6,000 IU (n=4) or 12,000 IU (n=2), administered orally three times weekly for six weeks
Setting: Five subjects underwent full-mouth dental procedures prior to treatment; one subject received dental cleaning only
Clinical and Market Context
FCGS is a debilitating, immune-mediated condition characterized by chronic oral inflammation, pain, and reduced quality of life. Current management often relies on invasive dental extractions and symptomatic therapies, with variable outcomes. An effective, non-invasive immunomodulatory approach could represent a meaningful advancement in companion animal care.
The global pet dental health market size is projected to reach USD 12.70 billion by 2030, growing at a CAGR of 7.57% from 2025 to 2030, according to Grand View Research.
About Ainos, Inc.
Ainos, Inc. (NASDAQ:AIMD) is a dual-platform AI and biotech company pioneering smelltech and immune therapeutics. Its AI Nose platform and smell language model (SLM) digitize scent into Smell ID, a machine-readable data format, powering intelligent sensing across robotics, smart factories, and healthcare. The company also develops VELDONA®, a low-dose oral interferon targeting rare, autoimmune, and infectious diseases. Ainos, a fusion of "AI" and "Nose," is redefining machine perception for the sensory age. To learn more, visit https://www.ainos.com. Follow Ainos on X, formerly known as Twitter, (@AinosInc) and LinkedIn to stay up-to-date.
Forward-Looking Statements
Certain statements in this press release are forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. All statements other than statements of historical fact are forward-looking statements. Forward-looking statements are based on management's current assumptions and expectations of future events and trends, which affect or may affect the Company's business, strategy, operations or financial performance, and actual results and other events may differ materially from those expressed or implied in such statements due to numerous risks and uncertainties. Forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified. There are a number of important factors that could cause actual results, developments, business decisions or other events to differ materially from those contemplated by the forward-looking statements in this press release. These factors include, among other things, our expectation that we will incur net losses for the foreseeable future; our ability to become profitable; our ability to raise additional capital to continue our product development; our ability to accurately predict our future operating results; our ability to advance our current or future product candidates through clinical trials, obtain marketing approval and ultimately commercialize any product candidates we develop; the ability to obtain and maintain regulatory approval of our product candidates; delays in completing the development and commercialization of our current and future product candidates; developing and commercializing additional products, including diagnostic testing devices; our ability to compete in the marketplace; compliance with applicable laws, regulations and tariffs, and factors described in the Risk Factors section of our public filings with the Securities and Exchange Commission (SEC). Because forward-looking statements are inherently subject to risks and uncertainties, you should not rely on these forward-looking statements as predictions of future events. These forward-looking statements speak only as of the date of this press release and, except to the extent required by applicable law, the Company undertakes no obligation to update or revise these statements, whether as a result of any new information, future events and developments or otherwise.
Investor Relations Contact
Feifei Shen
Email: [email protected]
SOURCE: Ainos, Inc.
View the original press release on ACCESS Newswire
Y.Baker--AT
