
-
 Swede goes on trial for pressuring wife to sell sex
Swede goes on trial for pressuring wife to sell sex
-
US inflation surges 3.3% as Iran war impact bites

-
 Vance warns Iran not to 'play' US at talks in Pakistan
Vance warns Iran not to 'play' US at talks in Pakistan
-
Fernandez remains out despite apology: Chelsea boss Rosenior

-
 Dortmund defender Schlotterbeck extends contract until 2031
Dortmund defender Schlotterbeck extends contract until 2031
-
De Zerbi vows to save troubled Spurs from relegation

-
 Antwerp port reopens to North Sea shipping after oil spill
Antwerp port reopens to North Sea shipping after oil spill
-
Stocks mixed, oil steadies on guarded optimism for Iran ceasefire

-
 Sinner eases into Monte Carlo semi-finals
Sinner eases into Monte Carlo semi-finals
-
France's Macron talks war, peace and basketball with Pope Leo

-
 Fernandez apologised over comments about his future: Chelsea's Rosenior
Fernandez apologised over comments about his future: Chelsea's Rosenior
-
Coach Spalletti signs new Juve deal until 2028

-
 AI chatbots offer children harm as if it were help, says activist
AI chatbots offer children harm as if it were help, says activist
-
'Grumpy' Guardiola wants Silva to stay at Man City for life

-
 Zverev beats Fonseca to reach Monte Carlo semi-finals
Zverev beats Fonseca to reach Monte Carlo semi-finals
-
Scheffler, Rose to chase McIlroy with early Masters starts

-
 Celine Dion's Paris concerts promise to spin the money on and on
Celine Dion's Paris concerts promise to spin the money on and on
-
Stocks climb, oil steadies on guarded optimism over Iran war ceasefire

-
 Irish govt to meet farmers, hauliers over fuel cost fears
Irish govt to meet farmers, hauliers over fuel cost fears
-
Injured Bayern starlet Karl to miss Real return leg

-
 US-Iran talks in Pakistan uncertain as sides trade accusations
US-Iran talks in Pakistan uncertain as sides trade accusations
-
Oil spill snarls shipping traffic in Antwerp port

-
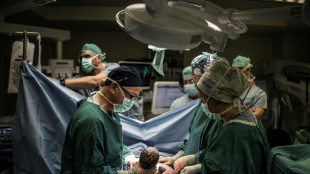 Giving birth in a shelter in Israel
Giving birth in a shelter in Israel
-
Five things to know about the planned Iran-US talks in Islamabad

-
 Slot feels 'complete support' from Liverpool chiefs despite slump
Slot feels 'complete support' from Liverpool chiefs despite slump
-
Kyiv books tentative diplomatic coup with Iran war forays

-
 Teenager shines as Britain seize control of BJK Cup tie with Australia
Teenager shines as Britain seize control of BJK Cup tie with Australia
-
Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader

-
 Sleepy seal diverts traffic in Australian seaside town
Sleepy seal diverts traffic in Australian seaside town
-
Artemis astronauts to shed light on space health risks

-
 Pakistan prepares to host US-Iran talks, as Lebanon fighting continues
Pakistan prepares to host US-Iran talks, as Lebanon fighting continues
-
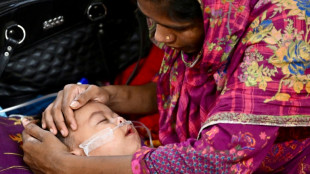
Vaccine gaps fuel Bangladesh's deadly measles crisis
-
 Fish furore fuels fierce election in India's West Bengal
Fish furore fuels fierce election in India's West Bengal
-
Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G

-
 Myanmar junta chief sworn in as president
Myanmar junta chief sworn in as president
-
Exiled cartoonists give voice to Iran's silenced millions

-
 In Pakistan's mediation to end Mideast war, China may hold the key
In Pakistan's mediation to end Mideast war, China may hold the key
-
Knicks stay in hunt with late win over rival Celtics

-
 'Sartorial diplomacy' on show in expo of late UK queen's fashion
'Sartorial diplomacy' on show in expo of late UK queen's fashion
-
Former Japan and AC Milan star Honda laces up boots again at 39

-
 Stocks rally on optimism over Iran war ceasefire, oil extends gains
Stocks rally on optimism over Iran war ceasefire, oil extends gains
-
Lego-style memes troll Trump after fragile US-Iran truce

-
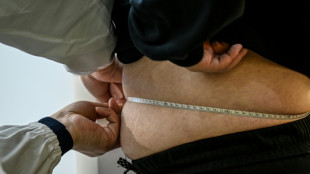 Chinese slimmers trade lost fat for beef
Chinese slimmers trade lost fat for beef
-
Jackson biopic shows franchise thriving despite abuse claims

-
 New Jersey city spurns data center as defiance spreads
New Jersey city spurns data center as defiance spreads
-
US box office looking good as cinema owners gather: industry chief

-
 Firm Masters greens make life hard on golf's finest
Firm Masters greens make life hard on golf's finest
-
BIO-key International, 20/20 Biolabs, Society Pass, and Soligenix Interviews to Air on the RedChip Small Stocks, Big Money(TM) Show on Bloomberg TV

-
 Designed for the Desk, Built for the Day: The Epomaker TH108 V2 Pro
Designed for the Desk, Built for the Day: The Epomaker TH108 V2 Pro
-
Releaf Crosses 25,000 Patients With Record Revenue


Xenetic Biosciences, Inc. Reports Second Quarter 2025 Financial Results
Expanded strategic partnership with The Scripps Research Institute to advance proof-of-concept studies and further develop its program combining systemic DNase I with CAR T-cell therapies
Ended the quarter with $4.8 million of cash to fund operations
FRAMINGHAM, MA / ACCESS Newswire / August 13, 2025 / Xenetic Biosciences, Inc. (NASDAQ:XBIO) ("Xenetic" or the "Company"), a biopharmaceutical company focused on advancing innovative immuno-oncology technologies addressing difficult to treat cancers, today reported its financial results for the second quarter 2025.
Recent Highlights
Expanded its collaboration with The Scripps Research Institute ("TSRI") to advance the development of the Company's development program evaluating the combination of systemic DNase I and CAR T-cell therapies;
Announced advancements from its collaboration partner, PeriNess Ltd. ("PeriNess") including:
Entered into a Clinical Study Agreement to support an exploratory clinical study of DNase I in combination with anti-CD19 CAR T cells in patients with large B cell lymphoma;
Commenced patient dosing in an exploratory clinical study of systemic DNase I in combination with FOLFIRINOX for the first line treatment of unresectable, locally advanced or metastatic pancreatic cancer at Bnei Zion Medical Center; and
Continued pursuit of other strategic collaborations to advance the Company's technology.
"We continue to set a strong foundation that we believe positions us for success as we advance our systemic DNase I in combination with immunotherapy, chemotherapy, and radiotherapy across various oncology indications where there remains significant unmet need. We continue to work with our partners and believe the data and information will be invaluable as we look to realize the full potential of our DNase platform technology. Looking ahead, we remain focused on building momentum across all fronts and driving development toward an IND and Phase 1 clinical trial," commented James Parslow, Interim Chief Executive Officer and Chief Financial Officer of Xenetic.
Xenetic continues to advance its DNase-based technology towards Phase 1 clinical development for the treatment of pancreatic carcinoma and other locally advanced or metastatic solid tumors. Preclinical proof-of-concept studies combining DNase I with chemotherapy, immunotherapies, and CAR-T therapy in hematological and solid tumor and metastatic cancer models have been completed. Building on proof-of-concept success, the program has now advanced to mechanism-of-action and translational studies in preparation for a Phase 1 clinical trial.
Additionally, as previously announced in December 2024, Xenetic entered into a Clinical Trial Services Agreement with PeriNess, under which PeriNess will lead in the regulatory approval, operational execution and management of potential exploratory, investigator initiated studies of recombinant DNase as an adjunctive treatment in patients with pancreatic carcinoma and other locally advanced or metastatic solid tumors receiving chemotherapy and immunotherapy in Israeli medical centers.
Summary of Financial Results for Second Quarter 2025
Net loss for the quarter ended June 30, 2025 was approximately $0.7 million. Research & development expenses for the three months ended June 30, 2025 decreased by approximately $277,000, or 29.7%, to approximately $0.7 million from $0.9 million in the comparable quarter in 2024. General and administrative expenses for the three months ended June 30, 2025 decreased by approximately $472,000, or 41.8%, to approximately $0.7 million from approximately $1.1 million in the comparable quarter in 2024. These decreases were primarily due to certain severance and benefits expensed in connection with separation agreements entered into during the second quarter of 2024 with the Company's former Chief Executive Officer and Chief Scientific Officer.
The Company ended the quarter with approximately $4.8 million in cash.
About Xenetic Biosciences
Xenetic Biosciences, Inc. is a biopharmaceutical company focused on advancing innovative immune-oncology technologies addressing hard to treat cancers. The Company's DNase platform is designed to improve outcomes of existing treatments, including immunotherapies, by targeting neutrophil extracellular traps (NETs), which are involved in cancer progression. Xenetic is currently focused on advancing its systemic DNase program into the clinic as an adjunctive therapy for pancreatic carcinoma and locally advanced or metastatic solid tumors.
For more information, please visit the Company's website at www.xeneticbio.com and connect on X, LinkedIn, and Facebook.
Forward-Looking Statements
This press release contains forward-looking statements that we intend to be subject to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release other than statements of historical facts may constitute forward-looking statements within the meaning of the federal securities laws. These statements can be identified by words such as "expects," "plans," "projects," "will," "may," "anticipates," "believes," "should," "intends," "estimates," "remain," "focus", "confidence in", "potential", and other words of similar meaning, including, but not limited to, all statements regarding expectations for our DNase-base oncology platform, including statements regarding: (a) advancing our DNase-based oncology program towards Phase 1 clinical development for the treatment of pancreatic carcinoma and other locally advanced or metastatic solid tumors and our focus on building momentum and driving development toward an IND and Phase I clinical trial, (b) setting a strong foundation that we believe positions us for success as we advance DNase I in combination with various types of therapy and (c) working with our partners and our belief regarding the data and information as we look to realize the full potential of DNase. Any forward-looking statements contained herein are based on current expectations and are subject to a number of risks and uncertainties. Many factors could cause our actual activities, performance, achievements, or results to differ materially from the activities and results anticipated in forward-looking statements. Important factors that could cause actual activities, performance, achievements, or results to differ materially from such plans, estimates or expectations include, among others, (1) unexpected costs, charges or expenses resulting from our manufacturing and collaboration agreements; (2) unexpected costs, charges or expenses resulting from the licensing of the DNase platform; (3) uncertainty of the expected financial performance of the Company following the licensing of the DNase platform; (4) failure to realize the anticipated potential of the DNase or PolyXen technologies; (5) the ability of the Company to obtain funding and implement its business strategy; and (6) other risk factors as detailed from time to time in the Company's reports filed with the SEC, including its annual report on Form 10-K, periodic quarterly reports on Form 10-Q, current reports on Form 8-K and other documents filed with the SEC. The foregoing list of important factors is not exclusive. In addition, forward-looking statements may also be adversely affected by general market factors, general economic and business conditions, including potential adverse effects of public health issues, and geopolitical events, such as the conflicts in Ukraine and in the Middle East, on economic activity, competitive product development, product availability, federal and state regulations and legislation, the regulatory process for new product candidates and indications, manufacturing issues that may arise, patent positions, litigation, and shareholder activism, among other factors. The forward-looking statements contained in this press release speak only as of the date the statements were made, and the Company does not undertake any obligation to update forward-looking statements, except as required by law.
Contact:
JTC Team, LLC
Jenene Thomas
(908) 824-0775
[email protected]
SOURCE: Xenetic Biosciences, Inc.
View the original press release on ACCESS Newswire
O.Ortiz--AT

