-
 Inter thump Roma to extend Serie A lead to nine points
Inter thump Roma to extend Serie A lead to nine points
-
Lebanon's Christians mark Easter in solidarity with war-hit south

-
 Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years
Leeds beat West Ham in shoot-out to reach FA Cup semis for first time in 39 years
-
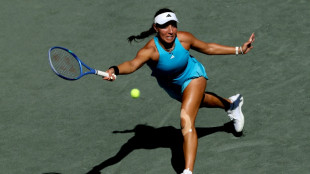
Pegula romps to WTA Charleston Open victory
-
 David six-hitting spree powers Bengaluru to IPL win
David six-hitting spree powers Bengaluru to IPL win
-
Union draw leaves St Pauli stranded in Bundesliga drop zone

-
 UK police arrest protesters near base used by US
UK police arrest protesters near base used by US
-
Trump issues foul-mouthed threat to Iran after US airman rescued

-
 Alcaraz plans to play full clay-court season, get 'socks dirty'
Alcaraz plans to play full clay-court season, get 'socks dirty'
-
'Super Mario Galaxy' blasts off in N. America box office debut

-
 Artemis astronauts begin fifth day on historic Moon mission
Artemis astronauts begin fifth day on historic Moon mission
-
Bielle-Biarrey sparkles as Bordeaux-Begles cruise in Champions Cup

-
 Trump draws criticism with fiery Easter message on Iran
Trump draws criticism with fiery Easter message on Iran
-
OPEC+ hikes oil production quotas, issues warning

-
 British PM slams London event for booking Kanye West, sponsor quits
British PM slams London event for booking Kanye West, sponsor quits
-
Pogacar wins joint-record third Tour of Flanders

-
 Trump threatens 'hell' for Iran over Strait of Hormuz
Trump threatens 'hell' for Iran over Strait of Hormuz
-
Shami, Pant help Lucknow beat Hyderabad in nervy IPL clash

-
 What we know about the race to rescue downed US airman in Iran
What we know about the race to rescue downed US airman in Iran
-
US commandos went deep into Iran to rescue downed airman: media

-
 Liberated McIlroy eyes more Masters magic after career Slam
Liberated McIlroy eyes more Masters magic after career Slam
-
Van Dijk apologises for Liverpool thumping by Man City

-
 British PM slams London festival for booking Kanye West
British PM slams London festival for booking Kanye West
-
'Choose peace': Pope marks first Easter under cloud of Mideast war

-
 British royals attend Easter service without Andrew
British royals attend Easter service without Andrew
-
US media says commandos probed deep into Iran to rescue downed airman

-
 Revellers parade giant penises to dash stigma in Japan's fertility festival
Revellers parade giant penises to dash stigma in Japan's fertility festival
-
Artemis astronauts glimpse Moon's 'Grand Canyon' ahead of historic lunar flyby

-
 Middle East war hits Britain's fish and chip shops
Middle East war hits Britain's fish and chip shops
-
Artemis astronauts to study the Moon's surface using mainly their eyes

-
 Second US airman downed over Iran 'SAFE and SOUND': Trump
Second US airman downed over Iran 'SAFE and SOUND': Trump
-
Indonesia lays to rest peacekeepers killed in Lebanon

-
 Pharmaceutical logistics in demand as war rattles supply chains
Pharmaceutical logistics in demand as war rattles supply chains
-
Messi marks new stadium with goal but Miami held by Austin

-
 Afghan mother seeks justice after Pakistani bombing kills hundreds
Afghan mother seeks justice after Pakistani bombing kills hundreds
-
UK royal family's dilemma over Andrew's daughters

-
 Pope marks first Easter under cloud of Mideast war
Pope marks first Easter under cloud of Mideast war
-
AI at war: Five things to know about Project Maven

-
 In the online 'maxxing' era, what's the deal with fiber and protein?
In the online 'maxxing' era, what's the deal with fiber and protein?
-
At Met Opera, life after a school shooting takes center stage

-
 Taiwan opposition leader to make 'peace' visit to China, first in 10 years
Taiwan opposition leader to make 'peace' visit to China, first in 10 years
-
McIlroy seeks rare Masters repeat in wide-open Augusta fight

-
 Israel says will strike Lebanon-Syria border crossing
Israel says will strike Lebanon-Syria border crossing
-
Global Energy Shift Accelerates: Surging Gas Prices Drive Mass EV Adoption - Elektros Advances Patented Technology Aimed at Transforming Charging Efficiency

-
 Paul topples Tiafoe to book Houston ATP final against Burruchaga
Paul topples Tiafoe to book Houston ATP final against Burruchaga
-
Jokic out-duels Wemby as Nuggets down Spurs in overtime

-
 Trump gives Iran 48 hours to make deal, search for missing airman continues
Trump gives Iran 48 hours to make deal, search for missing airman continues
-
Lens' title push in Ligue 1 hit hard by Lille defeat

-
 Arteta demands Arsenal response after FA Cup shocker at Southampton
Arteta demands Arsenal response after FA Cup shocker at Southampton
-
Barca move clear in La Liga as Real Madrid stumble


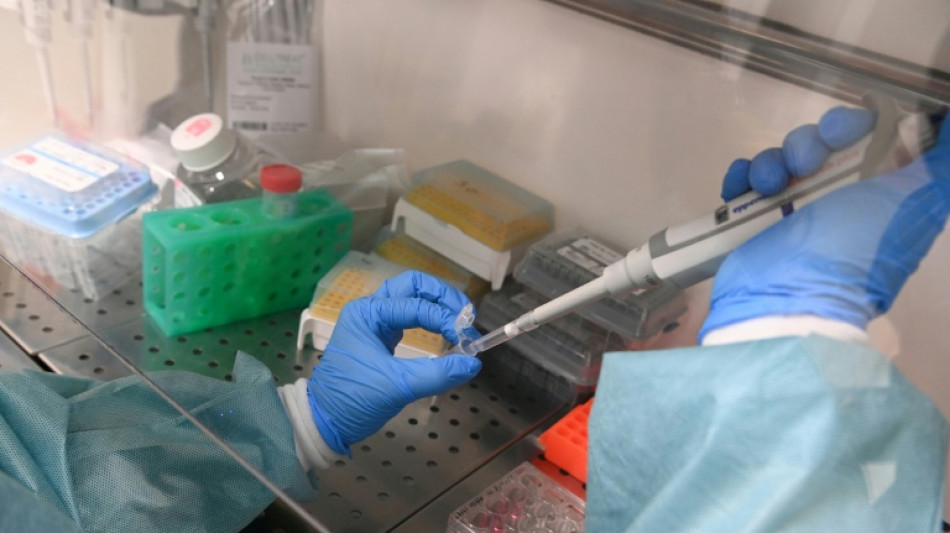
In reversal, US agrees to review new Moderna flu shot
Vaccine manufacturer Moderna said Wednesday the US Food and Drug Administration walked back its previous position and agreed to review the company's new mRNA-based flu shot.
Last week the US firm said the federal vaccine regulator rejected the application for review of the new shot, calling its clinical trial inadequate.
But Moderna said that after a "constructive" meeting the FDA had accepted the application for review based on a regulatory pathway focused on older adults.
The company's application now seeks full approval for adults 50 to 64 and accelerated approval for adults 65 and older, Moderna said in a statement. It also agreed to conduct an additional study after the shot hits the market.
The FDA's rejection had come as the body has called for a reconsideration of approval procedures for certain vaccines, including for influenza -- proposed federal policy changes under President Donald Trump that have triggered widespread alarm among public health and medical professionals.
The new shot uses mRNA technology, which health chief Robert F. Kennedy Jr, a vocal vaccine skeptic, has criticized. He notably cut off federal research grants that funded mRNA development.
That contradicts Trump's position during his first presidential term, when he called mRNA technology a "modern-day miracle."
It was used during the Covid-19 pandemic to swiftly develop an immunization that global health authorities deemed safe and effective against the fast-spreading illness. It was credited with saving millions of lives.
Moderna's new shot had already been accepted for review in the European Union, Canada and Australia.
Vinay Prasad, the top US vaccine official, had signed the letter rejecting Moderna's bid for approval, saying the company's clinical trial was not "adequate and well-controlled," and had not tested its experimental shot against the best product on the market.
In the large trial Moderna had compared its new vaccine with Fluarix, an approved flu shot from the company GSK.
Moderna said the rejection was "inconsistent with previous written communications" with the FDA.
In a statement Wednesday Moderna's CEO, Stephane Bancel, said "we appreciate the FDA's engagement" in a "constructive" meeting the company had requested following the rejection.
"Pending FDA approval, we look forward to making our flu vaccine available later this year so that America's seniors have access to a new option to protect themselves against flu."
Since Trump retook the White House, both he and health chief Kennedy have come under broad criticism from public health and medical experts for sowing doubt about the safety and efficacy of vaccines widely known to be safe, and upending the pediatric immunization schedule.
J.Gomez--AT

