
-
 Greece set new tourism record in 2025
Greece set new tourism record in 2025
-
Zelensky says Ukraine unbroken after 4 years, but Russia vows to fight on

-
 Zelenksy says Ukraine unbroken after 4 years, but Russia vows to fight on
Zelenksy says Ukraine unbroken after 4 years, but Russia vows to fight on
-
Snoop Dogg 'can't wait' for first Swansea visit

-
 Stocks fluctuate as traders assess AI fallout, tariffs
Stocks fluctuate as traders assess AI fallout, tariffs
-
Post-it maker 3M faces Belgian trial over 'forever' chemicals

-
 UK comedian Russell Brand pleads not guilty to new rape, assault charges
UK comedian Russell Brand pleads not guilty to new rape, assault charges
-
Duterte drew up 'death lists', boasted about murders: ICC prosecutor

-
 UK govt urged to release documents linked to ex-prince Andrew
UK govt urged to release documents linked to ex-prince Andrew
-
Rights group slams treatment of viral Japanese monkey

-
 Inside the bunker where Zelensky led response to Russian invasion
Inside the bunker where Zelensky led response to Russian invasion
-
France demands explanation from US envoy over 'surprise' no-show

-
 Putin failed to achieve goals in Ukraine, Zelensky says on war anniversary
Putin failed to achieve goals in Ukraine, Zelensky says on war anniversary
-
China tightens Japanese trade restrictions as spat worsens

-
 Ukraine war exhibition opens at Berlin Nazi bunker museum
Ukraine war exhibition opens at Berlin Nazi bunker museum
-
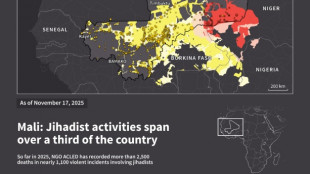
Jihadist threat puts eastern Senegal on edge
-
 Kim Yo Jong: the powerful sister behind North Korea's supreme leader
Kim Yo Jong: the powerful sister behind North Korea's supreme leader
-
North Korea ruling party promotes Kim Jong Un's younger sister

-
 Mexico's Jalisco cautiously tries returning to normal after cartel violence
Mexico's Jalisco cautiously tries returning to normal after cartel violence
-
Mexico's violence-hit Guadalajara to host World Cup games

-
 Mourinho's Bernabeu homecoming upended by suspension, racism row
Mourinho's Bernabeu homecoming upended by suspension, racism row
-
China targets Japanese companies over military ties

-
 Griezmann in talks to join MLS side Orlando City: source
Griezmann in talks to join MLS side Orlando City: source
-
France to revoke US envoy's govt access after summons no-show

-
 Spurs overpower Pistons in clash of NBA's form teams
Spurs overpower Pistons in clash of NBA's form teams
-
Inoue to fight Nakatani in Tokyo in May: reports

-
 Canada PM to push trade, rebuild fractured ties in India trip
Canada PM to push trade, rebuild fractured ties in India trip
-
Asian markets mixed as traders weigh AI and tariffs outlook

-
 Votes may 'melt like snow': Reform, Greens eye Labour UK bastion
Votes may 'melt like snow': Reform, Greens eye Labour UK bastion
-
Venezuela says exiles welcome to return following mass amnesty

-
 Australia buys parts for future AUKUS sub reactor
Australia buys parts for future AUKUS sub reactor
-
Ukraine marks four years since Russian invasion

-
 Brazil court to try politicians over hit on black councilwoman
Brazil court to try politicians over hit on black councilwoman
-
Interim president says Venezuelans welcome to return after amnesty law

-
 Man kills police officer in Moscow train station blast
Man kills police officer in Moscow train station blast
-
Despite drop in 2025, Russian oil exports exceed pre-war volumes: report

-
 ARIA Cybersecurity Announces Major Oil Refiner Deploys AZT PROTECT(TM)
ARIA Cybersecurity Announces Major Oil Refiner Deploys AZT PROTECT(TM)
-
Greene Concepts Announces Major Be Water Expansion in Walmart Stores Across the Southeast

-
 Fuse Battery Announces Amended Subscription Receipt Financing Details
Fuse Battery Announces Amended Subscription Receipt Financing Details
-
Lightwave Logic, Inc. Provides Update on Commercial Pipeline and Announces Timing of Fourth Quarter and Full Year 2025 Earnings Call

-
 Unlearn Advances Huntington's Disease AI Modeling Through Access to CHDI Foundation Data
Unlearn Advances Huntington's Disease AI Modeling Through Access to CHDI Foundation Data
-
Protagonist Therapeutics to Participate in Multiple Investment Bank Conferences in March 2026

-
 Specificity (OTCID:SPTY) to Present on the Emerging Growth Conference on February 26th, 2026.
Specificity (OTCID:SPTY) to Present on the Emerging Growth Conference on February 26th, 2026.
-
Havertys Reports Operating Results for Fourth Quarter 2025

-
 Viemed Healthcare Announces Year End 2025 Earnings Conference Call Details
Viemed Healthcare Announces Year End 2025 Earnings Conference Call Details
-
Galway Metals Drilling Intersects 9.0 g/t Gold Over 6.0m Beginning 15.0m from Surface at Southwest Deposit

-
 PeanutButterJelly Expands Affiliate Marketplace From 15 to 40 Affiliate Merchants; Website Sessions Rise 70%; Launches Conversion and Growth Optimization Plan
PeanutButterJelly Expands Affiliate Marketplace From 15 to 40 Affiliate Merchants; Website Sessions Rise 70%; Launches Conversion and Growth Optimization Plan
-
Digipower X Announces Uplisting to Cboe Canada

-
 Jaguar Mining Provides Update on Geologic Interpretation at the Chamé Gold Exploration Target, Brazil
Jaguar Mining Provides Update on Geologic Interpretation at the Chamé Gold Exploration Target, Brazil
-
Electrovaya Receives $10.5 Million P.O from Fortune 500 Customer


Amphastar Announces FDA Approval for Ipratropium Bromide HFA
RANCHO CUCAMONGA, CA / ACCESS Newswire / February 24, 2026 / Amphastar Pharmaceuticals, Inc. (NASDAQ:AMPH) announced that the U.S. Food and Drug Administration ("FDA") has approved the Company's Abbreviated New Drug Application ("ANDA") for Ipratropium Bromide HFA Inhalation Aerosol 17mcg/actuation. The FDA determined that Amphastar's Ipratropium Bromide HFA Inhalation Aerosol is bioequivalent and therapeutically equivalent to Boehringer Ingelheim's Atrovent® HFA Inhalation Aerosol.
Additionally, the FDA has confirmed that this product is eligible for 180-days of generic drug exclusivity for Ipratropium Bromide HFA Inhalation Aerosol as we were the first ANDA applicant with Paragraph IV certification. This exclusivity period will begin on the first day that Amphastar's Ipratropium Bromide HFA Inhalation Aerosol is commercially launched.
"We are excited to announce FDA approval of Ipratropium Bromide HFA Inhalation Aerosol, reinforcing the strength of our integrated R&D and manufacturing model and demonstrating our ability to deliver complex, high-value generics," said Dr. Jack Zhang, Amphastar's President and Chief Executive Officer. "We expect this launch to contribute meaningfully to our respiratory portfolio and to further strengthen our long-term growth strategy, capabilities and commitment to delivering impactful therapies to patients as we increase our efforts on the development of proprietary pipeline candidates."
Ipratropium is an anticholinergic indicated for the maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema. According to IQVIA, the U.S. sales for Atrovent® HFA were approximately $112 million for the 12 months ended December 31, 2025.
Amphastar plans to launch its Ipratropium Bromide HFA Inhalation Aerosol, previously referred to as AMP-007, early in the second quarter of 2026.
Pipeline Information
The Company currently has one ANDA and one biosimilar insulin filed with the FDA targeting products with a combined market size of over $1.7 billion, along with two biosimilar products in development targeting products with a market size exceeding $3.7 billion, and two generic products in development targeting products with a market size of over $1 billion. This market information is based on IQVIA data for the 12 months ended December 31, 2025. The Company is developing multiple proprietary products with injectable and intranasal dosage forms.
The Company's proprietary pipeline also includes four recently in-licensed products including three proprietary peptides targeting oncology and ophthalmology indications, and a fully synthetic corticotropin compound designed to address inflammatory and autoimmune conditions.
About Amphastar Pharmaceuticals, Inc.
Amphastar is a biopharmaceutical company that focuses on developing, manufacturing, and commercializing technically challenging generic and proprietary injectable, inhalation, and intranasal products. Additionally, the Company sells active pharmaceutical ingredient, or API products. Most of the Company's finished products are contracted and distributed through group purchasing organizations, drug wholesalers, and drug retailers. More information and resources are available at www.amphastar.com.
Amphastar's logo and other trademarks or service marks of Amphastar, including, but not limited to Amphastar ®, BAQSIMI ®, Primatene MIST ®, REXTOVY ®, Amphadase ®, AmpsilogTM and Cortrosyn ®, are the property of Amphastar.
Forward-Looking Statements
All statements in this press release referenced above that are not historical are forward-looking statements, including, among other things, statements relating to our expectations regarding future financial performance and business trends, our future growth and our ability to continue to scale, sales and marketing of our products, market size and expansion, product portfolio, product development, the timing of FDA filings or approvals, the timing of product launches, acquisitions and other matters related to our pipeline of product candidates, the timing and results of clinical trials, the impact of our products, including their potential for continued revenue growth, the strategic trajectory of and market for our product pipeline, our long-term strategic vision, our ability to leverage our existing expertise and technology, the impacts of any licensing agreements and ability to commercialize additional therapies, our in-house manufacturing expertise, our ability to deliver high-quality, affordable therapies to patients, our commercial momentum and position in the market. These statements are not facts but rather are based on Amphastar's historical performance and our current expectations, estimates, and projections regarding our business, operations, and other similar or related factors. Words such as "may," "might," "will," "could," "would," "should," "anticipate," "predict," "potential," "continue," "expect," "intend," "plan," "project," "believe," "estimate," and other similar or related expressions are used to identify these forward-looking statements, although not all forward-looking statements contain these words. You should not place undue reliance on forward-looking statements because they involve known and unknown risks, uncertainties, and assumptions that are difficult or impossible to predict and, in some cases, beyond Amphastar's control. Actual results may differ materially from those in the forward-looking statements as a result of a number of factors, including those described in Amphastar's filings with the Securities and Exchange Commission ("SEC"), including in our Annual Report on Form 10-K for the year ended December 31, 2024, filed with the SEC on March 3, 2025, in our Quarterly Report on Form 10-Q for the quarter ended March 31, 2025, filed with the SEC on May 8, 2025, in our Quarterly Report on Form 10-Q for the quarter ended June 30, 2025, filed with the SEC on August 7, 2025, in our Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, filed with the SEC on November 6, 2025, and our other filings or reports that we may file with the SEC. In particular, there can be no guarantee that our sales strategies will be successful, or that we will continue to experience significant sales of BAQSIMI ®. You can locate these reports through our website at http://ir.amphastar.com and on the SEC's website at www.sec.gov. The forward-looking statements in this release speak only as of the date of the release. Amphastar undertakes no obligation to revise or update information or any forward-looking statements in this press release referenced above to reflect events or circumstances in the future, even if new information becomes available or if subsequent events cause our expectations to change.
Contact Information:
Amphastar Pharmaceuticals, Inc.
Bill Peters
Chief Financial Officer
(909) 476-3416
SOURCE: Amphastar Pharmaceuticals, Inc.
View the original press release on ACCESS Newswire
Ch.Campbell--AT


