
-
 Knicks stay in hunt with late win over rival Celtics
Knicks stay in hunt with late win over rival Celtics
-
'Sartorial diplomacy' on show in expo of late UK queen's fashion

-
 Former Japan and AC Milan star Honda laces up boots again at 39
Former Japan and AC Milan star Honda laces up boots again at 39
-
Stocks rally on optimism over Iran war ceasefire, oil extends gains

-
 Lego-style memes troll Trump after fragile US-Iran truce
Lego-style memes troll Trump after fragile US-Iran truce
-
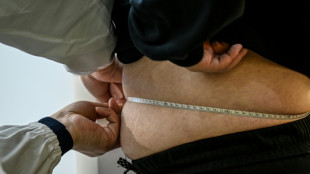
Chinese slimmers trade lost fat for beef
-
 Jackson biopic shows franchise thriving despite abuse claims
Jackson biopic shows franchise thriving despite abuse claims
-
New Jersey city spurns data center as defiance spreads

-
 US box office looking good as cinema owners gather: industry chief
US box office looking good as cinema owners gather: industry chief
-
Firm Masters greens make life hard on golf's finest

-
 Defending champ McIlroy shares Masters lead after back-nine birdie run
Defending champ McIlroy shares Masters lead after back-nine birdie run
-
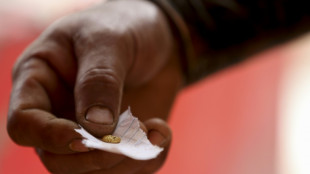
After oil, Venezuela opens up mining to private investors
-
 Tigers' Meadows in hospital after colliding with teammate
Tigers' Meadows in hospital after colliding with teammate
-
US to host Israel-Lebanon talks as strikes threaten Iran ceasefire

-
 'Scrappy' McIlroy leans on experience for share of Masters lead
'Scrappy' McIlroy leans on experience for share of Masters lead
-
Ukraine and Russia will cease fire for Orthodox Easter

-
 Mateta inspires Palace win over Fiorentina in Conference League
Mateta inspires Palace win over Fiorentina in Conference League
-
Pioneering US hip-hop artist Afrika Bambaataa dies at 68

-
 Russia bans Nobel-winning rights group, raids independent newspaper, in one day
Russia bans Nobel-winning rights group, raids independent newspaper, in one day
-
Pentagon denies giving Vatican envoy 'bitter lecture'

-
 Watkins propels Villa towards Europa League semis, Forest hold Porto
Watkins propels Villa towards Europa League semis, Forest hold Porto
-
Aston Villa on verge of Europa League semis after beating Bologna

-
 Venezuela police clash with protesters demanding salary rises
Venezuela police clash with protesters demanding salary rises
-
CAF president rejects corruption claims by Senegal

-
 Israel and Lebanon set for ceasefire talks next week, says US official
Israel and Lebanon set for ceasefire talks next week, says US official
-
US stocks extend gains, shrugging off ceasefire worries

-
 IMF chief urges nations to 'do no harm' in fiscal response to Iran war
IMF chief urges nations to 'do no harm' in fiscal response to Iran war
-
Sixers' Embiid to have surgery for appendicitis - team

-
 Russian police raid independent Novaya Gazeta outlet, reporter detained
Russian police raid independent Novaya Gazeta outlet, reporter detained
-
Former heavyweight king Fury adamant 'I've still got it' as Makhmudov awaits

-
 Shipping toll for Hormuz passage sharply divides nations
Shipping toll for Hormuz passage sharply divides nations
-
McIlroy's back-nine birdie run grabs share of Masters lead

-
 Melania Trump blasts 'lies' linking her to Epstein
Melania Trump blasts 'lies' linking her to Epstein
-
'Anxious' Tatum back at Madison Square Garden with NBA East second seed on line

-
 Strait of Hormuz traffic remains becalmed despite ceasefire
Strait of Hormuz traffic remains becalmed despite ceasefire
-
Melania Trump denies any links to Epstein abuse

-
 American Airlines targets April 30 return to Venezuela
American Airlines targets April 30 return to Venezuela
-
Venezuela police tear-gas protesters demanding salary rises

-
 Robertson to leave Liverpool at end of season
Robertson to leave Liverpool at end of season
-
Choudhary smashes Lucknow to dramatic IPL win over Kolkata

-
 Sean 'Diddy' Combs asks US appeals court to overturn sentence
Sean 'Diddy' Combs asks US appeals court to overturn sentence
-
Verstappen Red Bull future in doubt as engineer to join McLaren

-
 France's Macron in Rome for first meeting with Pope Leo
France's Macron in Rome for first meeting with Pope Leo
-
Angola name former Senegal boss Cisse as new coach

-
 Sinner and Alcaraz wobble but advance to Monte Carlo quarter-finals
Sinner and Alcaraz wobble but advance to Monte Carlo quarter-finals
-
Reed soars to early Masters lead on wings of eagles

-
 US Democrats fail in bid to curb Trump's Iran war powers
US Democrats fail in bid to curb Trump's Iran war powers
-
Veteran prop Slimani to return to France with Toulon

-
 Iranians pay tribute to slain supreme leader weeks after killing
Iranians pay tribute to slain supreme leader weeks after killing
-
Russian police raid independent Novaya Gazeta media outlet


EMA Committee for Medicinal Products for Human Use Adopts Positive Opinion Recommending Authorization of Moderna's mNEXSPIKE (COVID-19 Vaccine, mRNA)
mNEXSPIKE is Moderna's third product to receive a positive CHMP opinion alongside Spikevax (COVID-19 Vaccine, mRNA) and mRESVIA (Respiratory Syncytial Virus Vaccine)
mNEXSPIKE will be available in the European Union, subject to authorization by the European Commission
CAMBRIDGE, MA / ACCESS Newswire / December 15, 2025 / Moderna, Inc. (NASDAQ:MRNA) today announced that the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion recommending marketing authorization of mNEXSPIKE® (mRNA-1283), a new COVID vaccine, for active immunization to prevent COVID-19 caused by SARS-CoV-2 in individuals aged 12 years and older. Following the CHMP's positive opinion, the European Commission will make a marketing authorization decision.
The CHMP's positive opinion for mNEXSPIKE reflects Moderna's continued commitment to advancing innovative vaccines that protect the most vulnerable," said Stéphane Bancel, Chief Executive Officer of Moderna. "Across Europe, adults aged 65 and older remain disproportionately affected by severe COVID outcomes, and if authorized, mNEXSPIKE will be another important tool in addressing this ongoing public health need. Europe represents a significant market for respiratory virus vaccines, and we are excited about the opportunity to drive sales growth in 2026 and 2027 through this potential new product launch and geographic diversification. I want to thank our teams for their exceptional work and the EMA for its thorough assessment."
Across Europe, COVID-19 continues to persist year-round, with periodic case increases driven by waning immunity and the emergence of new Omicron-lineage variants, as demonstrated through the European Centre for Disease Prevention and Control's (ECDC) respiratory virus surveillance system.[1] These trends confirm that COVID-19 has become an established, endemic respiratory pathogen in the region, requiring targeted protection for vulnerable populations.
The CHMP's positive opinion for mNEXSPIKE is supported by results from a randomized, observer-blind, active-controlled Phase 3 clinical trial (ClinicalTrials.gov Identifier: NCT05815498), which enrolled approximately 11,400 participants aged 12 years and older. The primary efficacy objective in this study was to demonstrate the non-inferior vaccine efficacy against COVID-19 starting 14 days after mNEXSPIKE compared to that after the comparator vaccine, mRNA-1273 (Spikevax®), Moderna's original COVID-19 vaccine. Participants received either a 10 μg dose of mRNA-1283 or a 50 μg dose of mRNA-1273. mRNA-1283 showed a 9.3% higher relative vaccine efficacy (rVE) compared to mRNA-1273 in individuals aged 12 years and older, and in a descriptive sub-group analysis, a 13.5% higher rVE in adults aged 65 and older.
In the Phase 3 trial, mRNA-1283 was found to have a similar safety profile to mRNA-1273, with fewer local reactions and comparable systemic reactions. The most commonly solicited side effects were injection site pain, fatigue, headache and myalgia.
Subject to the European Commission's decision, Moderna expects to make mNEXSPIKE available in Europe pending regulatory timelines and local market access pathways. mNEXSPIKE is Moderna's third product to receive a positive CHMP opinion alongside Spikevax and mRESVIA®, the Company's approved respiratory syncytial virus (RSV) vaccine.
Moderna has filed mRNA-1283 for approval with regulators in multiple markets around the world, and has received approval from the U.S. Food and Drug Administration (FDA) and Health Canada.
About Moderna
Moderna is a pioneer and leader in the field of mRNA medicine. Through the advancement of its technology platform, Moderna is reimagining how medicines are made to transform how we treat and prevent diseases. Since its founding, Moderna's mRNA platform has enabled the development of vaccines and therapeutics across infectious diseases, cancer, rare diseases and more.
With a global team and a unique culture, driven by the company's values and mindsets, Moderna's mission is to deliver the greatest possible impact to people through mRNA medicines. For more information about Moderna, please visit modernatx.com and connect with us on X, Facebook, Instagram, YouTube and LinkedIn.
Spikevax®, mRESVIA® and mNEXSPIKE® are registered trademarks of Moderna.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including statements regarding: the availability of mNEXSPIKE in Europe for the 2026-2027 season; the European Commission's pending marketing authorization decision; the opportunity in the European respiratory virus vaccine market; the ability of Moderna's COVID vaccines to generate an immune response and to protect against circulating COVID strains; and the safety profile of mNEXSPIKE. The forward-looking statements in this press release are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond Moderna's control and which could cause actual results to differ materially from those expressed or implied by these forward-looking statements. These risks, uncertainties, and other factors include, among others, those risks and uncertainties described under the heading "Risk Factors" in Moderna's Annual Report on Form 10-K for the fiscal year ended December 31, 2024, and in subsequent filings made by Moderna with the U.S. Securities and Exchange Commission, which are available on the SEC's website at www.sec.gov. Except as required by law, Moderna disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this press release in the event of new information, future developments or otherwise. These forward-looking statements are based on Moderna's current expectations and speak only as of the date of this press release.
Moderna Contacts
Media:
Chris Ridley
Head of Global Media Relations
+1 617-800-3651
[email protected]
Investors:
Lavina Talukdar
Senior Vice President & Head of Investor Relations
+1 617-209-5834
[email protected]
SOURCE: Moderna, Inc.
View the original press release on ACCESS Newswire
T.Wright--AT

