-
 War's impact on fertilisers stirs food producer fears
War's impact on fertilisers stirs food producer fears
-
US inflation surges to 3.3% as Iran war impact bites

-
 Thais fete new year with family despite fuel price spike
Thais fete new year with family despite fuel price spike
-
Scheffler scrambles, Rose stumbles early at Masters

-
 On Iran truce, all sides want bigger China role, but does China?
On Iran truce, all sides want bigger China role, but does China?
-
Sinner eases into Monte Carlo semi-final against Zverev

-
 Inter skipper Martinez suffers calf injury
Inter skipper Martinez suffers calf injury
-
Ukrainians sceptical as Kremlin orders Easter truce

-
 Arteta urges Arsenal to pile pressure on Man City in title race
Arteta urges Arsenal to pile pressure on Man City in title race
-
Pay fears grow for US security workers in shutdown

-
 Hungary rivals rally crowds in closing strait of election campaign
Hungary rivals rally crowds in closing strait of election campaign
-
Swede goes on trial for pressuring wife to sell sex

-
 US inflation surges 3.3% as Iran war impact bites
US inflation surges 3.3% as Iran war impact bites
-
Vance warns Iran not to 'play' US at talks in Pakistan

-
 Fernandez remains out despite apology: Chelsea boss Rosenior
Fernandez remains out despite apology: Chelsea boss Rosenior
-
Dortmund defender Schlotterbeck extends contract until 2031

-
 De Zerbi vows to save troubled Spurs from relegation
De Zerbi vows to save troubled Spurs from relegation
-
Antwerp port reopens to North Sea shipping after oil spill

-
 Stocks mixed, oil steadies on guarded optimism for Iran ceasefire
Stocks mixed, oil steadies on guarded optimism for Iran ceasefire
-
Sinner eases into Monte Carlo semi-finals

-
 France's Macron talks war, peace and basketball with Pope Leo
France's Macron talks war, peace and basketball with Pope Leo
-
Fernandez apologised over comments about his future: Chelsea's Rosenior

-
 Coach Spalletti signs new Juve deal until 2028
Coach Spalletti signs new Juve deal until 2028
-
AI chatbots offer children harm as if it were help, says activist

-
 'Grumpy' Guardiola wants Silva to stay at Man City for life
'Grumpy' Guardiola wants Silva to stay at Man City for life
-
Zverev beats Fonseca to reach Monte Carlo semi-finals

-
 Scheffler, Rose to chase McIlroy with early Masters starts
Scheffler, Rose to chase McIlroy with early Masters starts
-
Celine Dion's Paris concerts promise to spin the money on and on

-
 Stocks climb, oil steadies on guarded optimism over Iran war ceasefire
Stocks climb, oil steadies on guarded optimism over Iran war ceasefire
-
Irish govt to meet farmers, hauliers over fuel cost fears

-
 Injured Bayern starlet Karl to miss Real return leg
Injured Bayern starlet Karl to miss Real return leg
-
US-Iran talks in Pakistan uncertain as sides trade accusations

-
 Oil spill snarls shipping traffic in Antwerp port
Oil spill snarls shipping traffic in Antwerp port
-
Giving birth in a shelter in Israel
-
 Five things to know about the planned Iran-US talks in Islamabad
Five things to know about the planned Iran-US talks in Islamabad
-
Slot feels 'complete support' from Liverpool chiefs despite slump

-
 Kyiv books tentative diplomatic coup with Iran war forays
Kyiv books tentative diplomatic coup with Iran war forays
-
Teenager shines as Britain seize control of BJK Cup tie with Australia

-
 Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader
Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader
-
Sleepy seal diverts traffic in Australian seaside town

-
 Artemis astronauts to shed light on space health risks
Artemis astronauts to shed light on space health risks
-
Pakistan prepares to host US-Iran talks, as Lebanon fighting continues

-
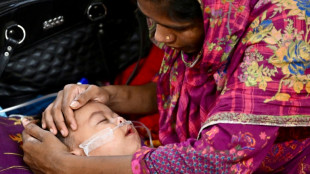 Vaccine gaps fuel Bangladesh's deadly measles crisis
Vaccine gaps fuel Bangladesh's deadly measles crisis
-
Fish furore fuels fierce election in India's West Bengal

-
 Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G
Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G
-
Myanmar junta chief sworn in as president

-
 Exiled cartoonists give voice to Iran's silenced millions
Exiled cartoonists give voice to Iran's silenced millions
-
In Pakistan's mediation to end Mideast war, China may hold the key

-
 Knicks stay in hunt with late win over rival Celtics
Knicks stay in hunt with late win over rival Celtics
-
'Sartorial diplomacy' on show in expo of late UK queen's fashion


President Trump Fixing DEA Marijuana Policy: Defies Logic, Law, and Science, Says MMJ International Holdings
MMJ International Holdings Exposes the Impossible Federal Catch-22 Blocking FDA Regulated Cannabis Medicine. "This is an equation with no solution. To follow the rule is to commit a felony, and to follow the law is to violate the rule. This is the first paradox I've encountered that violates logic and the U.S. criminal code simultaneously." stated Duane Boise, CEO MMJ International Holdings.
WASHINGTON, D.C. / ACCESS Newswire / December 3, 2025 / MMJ International Holdings, a pharmaceutical developer of a fully federally compliant, natural cannabinoid pharmaceutical for Huntington's Disease and Multiple Sclerosis, today released a sharply critical analysis of the Drug Enforcement Administration's marijuana research policies-calling them "mathematically impossible, legally contradictory, and scientifically indefensible."

FDA Orphan Drug Designation
FDA IND programs prepared for Phase 2/3 trials
Fully manufactured GMP softgel capsules
Completed stability data
A DEA Schedule I import authorization
The barrier is not science or safety-but DEA procedure.
A Regulatory Paradox With No Legal Route Forward
To obtain a DEA bulk manufacturing registration for pharmaceutical cannabis, the agency requires applicants to first present a bona fide supply contract with another federally licensed cannabis manufacturer.
But federal law classifies cannabis as a Schedule I drug, making any negotiation, drafting, or agreement involving future cannabis supply a federal trafficking conspiracy.
Meaning:
It is illegal to negotiate the contract DEA demands, and
It is impossible to obtain the license without it.
MMJ International Holdings CEO Duane Boise described the situation plainly:
"This is a regulatory equation with no solution.
To follow the rule is to commit a felony, and to follow the law is to violate the rule.
DEA has created a system where compliance itself is illegal."
Meanwhile, Ultra-Potent THC Products Flourish in Plain Sight
While federally compliant pharmaceutical developers are blocked, state dispensaries nationwide openly sell high-potency THC concentrates, many reaching high levels of THC. These products:
Remain federally illegal under the Controlled Substances Act
Are linked by peer-reviewed research to psychosis, schizophrenia, and neurological destabilization
Are widely accessible to young adults
Yet MMJ-operating entirely under federal authority-still cannot secure approval for:
Consistent, stabilized pharmaceutical softgel capsules
Produced under GMP standards
Using natural full-spectrum cannabis extracts
Intended exclusively for FDA-supervised human clinical trials
Boise added:
A System Officially Declared Unconstitutional
In 2025, the U.S. Department of Justice formally acknowledged that the DEA's Administrative Law Judge (ALJ) system-the tribunal required to adjudicate MMJ's case-was "unconstitutional from the start."
As a result:
MMJ never received a lawful evidentiary hearing
The company's 2018 application remains in limbo
There is no functional mechanism to resolve the case without decisive administrative action
Boise characterized the situation as:
"Seven years of delay, despite full compliance, fully manufactured medicine, and FDA readiness.
This is not regulation-it is regulatory entropy."
The Human Cost Behind the Bureaucracy
The satire underscores a deadly serious problem:
Huntington's Disease patients experience chorea-violent involuntary movements that destroy independence and quality of life
MMJ's treatment has FDA Orphan Drug Designation
The medicine is manufactured and ready
The only barrier is one agency's procedural loop
Boise stressed:
"Every day of DEA delay is another day patients go without hope.
We built real medicine-standardized, federally legal medicine-yet we are blocked while dangerous products flourish unchecked."
MMJ Calls for Immediate Federal Action
MMJ International Holdings urges the federal government and DEA Administrator Terrance Cole to:
Resolve MMJ's application immediately under the Administrative Procedure Act
End the Catch-22 that makes compliance legally impossible
Prioritize pharmaceutical-grade cannabinoid medicine over unregulated high-potency THC
Align federal policy with science, public health, and international treaty obligations
Boise concluded:
"The universe is complicated-federal cannabis policy doesn't have to be.
Medicine should not be illegal while illegality is treated as medicine."
About MMJ International Holdings
MMJ International Holdings is a U.S.-based pharmaceutical company developing FDA-regulated, natural cannabinoid medicines. MMJ's subsidiaries-MMJ BioPharma Labs and MMJ BioPharma Cultivation-hold federal designations, IND programs, and DEA licensure for pharmaceutical research targeting Huntington's Disease and Multiple Sclerosis.
MMJ is represented by attorney Megan Sheehan.
CONTACT:
Madison Hisey
[email protected]
203-231-8583
SOURCE: MMJ International Holdings
View the original press release on ACCESS Newswire
M.O.Allen--AT