
-
 Trump says Iran has 'no cards' beyond Hormuz control
Trump says Iran has 'no cards' beyond Hormuz control
-
Israeli strike in south Lebanon kills 13 security personnel

-
 Will The Wise wins Topham as tragedy strikes Gold Dancer
Will The Wise wins Topham as tragedy strikes Gold Dancer
-
Over 100,000 worshippers perform Friday prayers at Al-Aqsa

-
 Teen star Seixas claims stage five to close on Basque Tour victory
Teen star Seixas claims stage five to close on Basque Tour victory
-
War's impact on fertilisers stirs food producer fears

-
 US inflation surges to 3.3% as Iran war impact bites
US inflation surges to 3.3% as Iran war impact bites
-
Thais fete new year with family despite fuel price spike

-
 Scheffler scrambles, Rose stumbles early at Masters
Scheffler scrambles, Rose stumbles early at Masters
-
On Iran truce, all sides want bigger China role, but does China?

-
 Sinner eases into Monte Carlo semi-final against Zverev
Sinner eases into Monte Carlo semi-final against Zverev
-
Inter skipper Martinez suffers calf injury

-
 Ukrainians sceptical as Kremlin orders Easter truce
Ukrainians sceptical as Kremlin orders Easter truce
-
Arteta urges Arsenal to pile pressure on Man City in title race

-
 Pay fears grow for US security workers in shutdown
Pay fears grow for US security workers in shutdown
-
Hungary rivals rally crowds in closing strait of election campaign

-
 Swede goes on trial for pressuring wife to sell sex
Swede goes on trial for pressuring wife to sell sex
-
US inflation surges 3.3% as Iran war impact bites

-
 Vance warns Iran not to 'play' US at talks in Pakistan
Vance warns Iran not to 'play' US at talks in Pakistan
-
Fernandez remains out despite apology: Chelsea boss Rosenior

-
 Dortmund defender Schlotterbeck extends contract until 2031
Dortmund defender Schlotterbeck extends contract until 2031
-
De Zerbi vows to save troubled Spurs from relegation

-
 Antwerp port reopens to North Sea shipping after oil spill
Antwerp port reopens to North Sea shipping after oil spill
-
Stocks mixed, oil steadies on guarded optimism for Iran ceasefire

-
 Sinner eases into Monte Carlo semi-finals
Sinner eases into Monte Carlo semi-finals
-
France's Macron talks war, peace and basketball with Pope Leo

-
 Fernandez apologised over comments about his future: Chelsea's Rosenior
Fernandez apologised over comments about his future: Chelsea's Rosenior
-
Coach Spalletti signs new Juve deal until 2028

-
 AI chatbots offer children harm as if it were help, says activist
AI chatbots offer children harm as if it were help, says activist
-
'Grumpy' Guardiola wants Silva to stay at Man City for life

-
 Zverev beats Fonseca to reach Monte Carlo semi-finals
Zverev beats Fonseca to reach Monte Carlo semi-finals
-
Scheffler, Rose to chase McIlroy with early Masters starts

-
 Celine Dion's Paris concerts promise to spin the money on and on
Celine Dion's Paris concerts promise to spin the money on and on
-
Stocks climb, oil steadies on guarded optimism over Iran war ceasefire

-
 Irish govt to meet farmers, hauliers over fuel cost fears
Irish govt to meet farmers, hauliers over fuel cost fears
-
Injured Bayern starlet Karl to miss Real return leg

-
 US-Iran talks in Pakistan uncertain as sides trade accusations
US-Iran talks in Pakistan uncertain as sides trade accusations
-
Oil spill snarls shipping traffic in Antwerp port

-
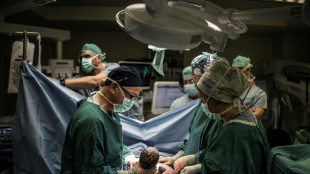 Giving birth in a shelter in Israel
Giving birth in a shelter in Israel
-
Five things to know about the planned Iran-US talks in Islamabad

-
 Slot feels 'complete support' from Liverpool chiefs despite slump
Slot feels 'complete support' from Liverpool chiefs despite slump
-
Kyiv books tentative diplomatic coup with Iran war forays

-
 Teenager shines as Britain seize control of BJK Cup tie with Australia
Teenager shines as Britain seize control of BJK Cup tie with Australia
-
Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader

-
 Sleepy seal diverts traffic in Australian seaside town
Sleepy seal diverts traffic in Australian seaside town
-
Artemis astronauts to shed light on space health risks

-
 Pakistan prepares to host US-Iran talks, as Lebanon fighting continues
Pakistan prepares to host US-Iran talks, as Lebanon fighting continues
-
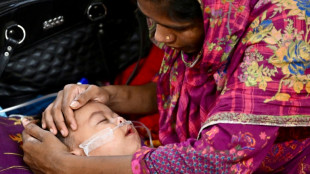
Vaccine gaps fuel Bangladesh's deadly measles crisis
-
 Fish furore fuels fierce election in India's West Bengal
Fish furore fuels fierce election in India's West Bengal
-
Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G


Ensysce Biosciences Announces Broader Patent Protection for Groundbreaking MPAR(R) Overdose Protection Technology
~ New U.S. Patent Extends Protection Through 2042 ~
~ Patent Allowance Secures Highest Level of Intellectual Property Protection for MPAR® ~
SAN DIEGO, CA / ACCESS Newswire / December 2, 2025 / Ensysce Biosciences, Inc. (NASDAQ:ENSC) ("Ensysce" or the "Company"), a clinical-stage pharmaceutical company pioneering next-generation pain and central nervous system therapeutics designed to minimize abuse and overdose risk, today announced that the U.S. Patent and Trademark Office has issued a Notice of Allowance for a new patent covering Ensysce's MPAR® (Multi-Pill Abuse Resistance) technology. The patent, titled "Compositions Comprising Enzyme-Cleavable Prodrugs and Controlled Release Nafamostat and Methods of Use Thereof", includes both composition-of-matter and method-of-use claims that strengthen Ensysce's intellectual property estate for its unique overdose protection platform. The new protection extends the Company's MPAR® patent coverage through 2042.
MPAR®, which earned Breakthrough Therapy Designation from the U.S. Food and Drug Administration (FDA) in January 2025, is designed to limit the effects of opioid exposure in the event of an overdose. In clinical testing, PF614-MPAR demonstrated effective pain relief when taken as prescribed, while preventing excessive opioid delivery when multiple doses were administered. Beyond opioids, Ensysce is applying its MPAR® technology to additional drug classes, including amphetamines and methadone, to develop safer treatments for pain, ADHD, and opioid use disorder.
"This new patent represents another important milestone in our mission to make vital and proven medicines safer," said Dr. Lynn Kirkpatrick, Chief Executive Officer of Ensysce. "MPAR® overdose protection, in combination with our TAAP™ abuse-deterrent platform, delivers a dual safety approach which we believe is unlike anything else in the field. We are grateful for support and funding from the National Institute on Drug Abuse1 that has allowed us to demonstrate clinically that MPAR moderates opioid release when taken in excess. The second clinical trial to further evaluate the potential of this technology is ongoing. We are striving to advance both PF614 and PF614-MPAR through late clinical development to achieve our goal of reducing opioid abuse and overdose."
¹Research supporting this patent was funded by the National Institute on Drug Abuse of the National Institutes of Health under Award Number DA047682.
About Ensysce Biosciences
Ensysce Biosciences is a clinical-stage company with a goal of disrupting the analgesic landscape by introducing a new class of highly novel opioids for the treatment of severe pain. Leveraging its Trypsin-Activated Abuse Protection (TAAP™) and Multi-Pill Abuse Resistance (MPAR®) platforms, the Company is developing unique, tamper-proof treatment options for pain that minimize the risk of both drug abuse and overdose. Ensysce's products are anticipated to provide safer options to treat patients suffering from severe pain and assist in preventing deaths caused by medication abuse. For more information, please visit www.ensysce.com.
Forward-Looking Statements
Statements contained in this press release that are not purely historical may be deemed to be forward-looking statements for the purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995 and other federal securities laws. Without limiting the foregoing, the use of words such as "may," "intends," "can," "might," "will," "expect," "plan," "possible," "believe" and other similar expressions are intended to identify forward-looking statements. The product candidates discussed are in clinic and not approved and there can be no assurance that the clinical programs will be successful in demonstrating safety and/or efficacy, that Ensysce will not encounter problems or delays in clinical development, or that any product candidate will ever receive regulatory approval or be successfully commercialized. All forward-looking statements are based on estimates and assumptions by Ensysce's management that, although Ensysce believes to be reasonable, are inherently uncertain. All forward-looking statements are subject to risks and uncertainties that may cause actual results to differ materially from those that Ensysce expected. In addition, Ensysce's business is subject to additional risks and uncertainties, including among others, possible NASDAQ delisting, the initiation and conduct of preclinical studies and clinical trials; the timing and availability of data from preclinical studies and clinical trials; expectations for regulatory submissions and approvals; potential safety concerns related to, or efficacy of, Ensysce's product candidates; the availability or commercial potential of product candidates; continuation of government funding; the ability of Ensysce to fund its continued operations, including its planned clinical trials; the dilutive effect of stock issuances from our fundraising; and Ensysce's and its partners' ability to perform under their license, collaboration and manufacturing arrangements. These statements are also subject to a number of material risks and uncertainties that are described in Ensysce's most recent quarterly report on Form 10-Q and current reports on Form 8-K, available free of charge at the SEC's website at www.sec.gov. Any forward-looking statement speaks only as of the date on which it was made. Ensysce undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required under applicable law.
Ensysce Biosciences Company Contact:
Lynn Kirkpatrick, Ph.D.
Chief Executive Officer
(858) 263-4196
Ensysce Biosciences Investor Relations Contact:
Shannon Devine
MZ North America
Main: 203-741-8811
[email protected]
SOURCE: Ensysce Biosciences
View the original press release on ACCESS Newswire
T.Perez--AT

