
-
 Trump says Iran has 'no cards' beyond Hormuz control
Trump says Iran has 'no cards' beyond Hormuz control
-
Israeli strike in south Lebanon kills 13 security personnel

-
 Will The Wise wins Topham as tragedy strikes Gold Dancer
Will The Wise wins Topham as tragedy strikes Gold Dancer
-
Over 100,000 worshippers perform Friday prayers at Al-Aqsa

-
 Teen star Seixas claims stage five to close on Basque Tour victory
Teen star Seixas claims stage five to close on Basque Tour victory
-
War's impact on fertilisers stirs food producer fears

-
 US inflation surges to 3.3% as Iran war impact bites
US inflation surges to 3.3% as Iran war impact bites
-
Thais fete new year with family despite fuel price spike

-
 Scheffler scrambles, Rose stumbles early at Masters
Scheffler scrambles, Rose stumbles early at Masters
-
On Iran truce, all sides want bigger China role, but does China?

-
 Sinner eases into Monte Carlo semi-final against Zverev
Sinner eases into Monte Carlo semi-final against Zverev
-
Inter skipper Martinez suffers calf injury

-
 Ukrainians sceptical as Kremlin orders Easter truce
Ukrainians sceptical as Kremlin orders Easter truce
-
Arteta urges Arsenal to pile pressure on Man City in title race

-
 Pay fears grow for US security workers in shutdown
Pay fears grow for US security workers in shutdown
-
Hungary rivals rally crowds in closing strait of election campaign

-
 Swede goes on trial for pressuring wife to sell sex
Swede goes on trial for pressuring wife to sell sex
-
US inflation surges 3.3% as Iran war impact bites

-
 Vance warns Iran not to 'play' US at talks in Pakistan
Vance warns Iran not to 'play' US at talks in Pakistan
-
Fernandez remains out despite apology: Chelsea boss Rosenior

-
 Dortmund defender Schlotterbeck extends contract until 2031
Dortmund defender Schlotterbeck extends contract until 2031
-
De Zerbi vows to save troubled Spurs from relegation

-
 Antwerp port reopens to North Sea shipping after oil spill
Antwerp port reopens to North Sea shipping after oil spill
-
Stocks mixed, oil steadies on guarded optimism for Iran ceasefire

-
 Sinner eases into Monte Carlo semi-finals
Sinner eases into Monte Carlo semi-finals
-
France's Macron talks war, peace and basketball with Pope Leo

-
 Fernandez apologised over comments about his future: Chelsea's Rosenior
Fernandez apologised over comments about his future: Chelsea's Rosenior
-
Coach Spalletti signs new Juve deal until 2028

-
 AI chatbots offer children harm as if it were help, says activist
AI chatbots offer children harm as if it were help, says activist
-
'Grumpy' Guardiola wants Silva to stay at Man City for life

-
 Zverev beats Fonseca to reach Monte Carlo semi-finals
Zverev beats Fonseca to reach Monte Carlo semi-finals
-
Scheffler, Rose to chase McIlroy with early Masters starts

-
 Celine Dion's Paris concerts promise to spin the money on and on
Celine Dion's Paris concerts promise to spin the money on and on
-
Stocks climb, oil steadies on guarded optimism over Iran war ceasefire

-
 Irish govt to meet farmers, hauliers over fuel cost fears
Irish govt to meet farmers, hauliers over fuel cost fears
-
Injured Bayern starlet Karl to miss Real return leg

-
 US-Iran talks in Pakistan uncertain as sides trade accusations
US-Iran talks in Pakistan uncertain as sides trade accusations
-
Oil spill snarls shipping traffic in Antwerp port

-
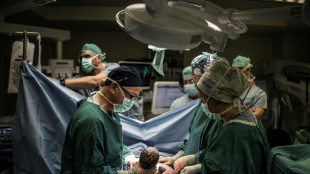 Giving birth in a shelter in Israel
Giving birth in a shelter in Israel
-
Five things to know about the planned Iran-US talks in Islamabad

-
 Slot feels 'complete support' from Liverpool chiefs despite slump
Slot feels 'complete support' from Liverpool chiefs despite slump
-
Kyiv books tentative diplomatic coup with Iran war forays

-
 Teenager shines as Britain seize control of BJK Cup tie with Australia
Teenager shines as Britain seize control of BJK Cup tie with Australia
-
Chinese, Taiwanese will unite, Xi tells Taiwan opposition leader

-
 Sleepy seal diverts traffic in Australian seaside town
Sleepy seal diverts traffic in Australian seaside town
-
Artemis astronauts to shed light on space health risks

-
 Pakistan prepares to host US-Iran talks, as Lebanon fighting continues
Pakistan prepares to host US-Iran talks, as Lebanon fighting continues
-
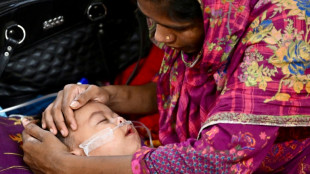
Vaccine gaps fuel Bangladesh's deadly measles crisis
-
 Fish furore fuels fierce election in India's West Bengal
Fish furore fuels fierce election in India's West Bengal
-
Coachella kicks off with headliners Sabrina Carpenter, Bieber and Karol G


GEN Announces New Positive Phase 1 Trial Data of the Investigational Drug SUL-238 for Alzheimer's and Other Neurodegenerative Diseases
Phase 1 results show that SUL-238 is safe, well-tolerated, and demonstrates favorable pharmacokinetics with high CSF penetration in healthy elderly volunteers, supporting its advancement into further clinical development for Alzheimer's and other neurodegenerative diseases.
ANKARA, TR / ACCESS Newswire / December 2, 2025 / GEN Pharmaceuticals (GENIL.IS), Türkiye's leading specialty pharmaceutical company, has announced new positive results from its Phase 1 clinical trial evaluating the safety, tolerability, and pharmacokinetics (PK) of first-in-class, novel orally administered mitochondria-directed drug candidate SUL-238 in healthy elderly volunteers. The findings were presented at the 18th Clinical Trials on Alzheimer's Disease (CTAD) in San Diego, CA (United States) today.
SUL-238 was originally discovered by Sulfateq and has since been further developed through a collaborative effort of Sulfateq and GEN as a novel therapeutic in neurodegenerative diseases.
This Phase 1 randomized, double-blind, placebo-controlled study evaluated the safety, tolerability, and pharmacokinetics (PK) after multiple-ascending doses (MAD) of orally administered SUL-238 in healthy elderly men and women (aged ≥40 years). The study included two cohorts with a treatment period of 14 days and a safety follow-up through 14 days after the last dose. 15 healthy adults in each cohort were randomized in a 2:1 ratio to receive SUL-238 or placebo. Total daily dose of SUL-238 was 4000 mg (2000 mg b.i.d., first cohort) or 4500 mg (1500 mg t.i.d., second cohort). SUL-238 demonstrated an excellent safety and tolerability profile after multiple doses in both cohorts, while demonstrating a favourable PK profile and a high cerebrospinal fluid (CSF) penetration, making it a promising candidate for further clinical development in neurodegenerative diseases, including Alzheimer's and Parkinson's diseases.
Key Findings:
Safety in both cohorts:
No clinically significant changes were observed in physical and neurological exams, vital signs, ECG, and clinical laboratory parameters.
AE rates were comparable between participants receiving SUL-238 and placebo.
All AEs were of mild intensity or considered not related to SUL-238.
First cohort PK (2000 mg b.i.d.):
SUL-238 was rapidly absorbed with a mean time to maximum plasma concentration (Tmax) reached at 1.25(±0.54) and 1.50(±0.53) hours on day 1 and day 14, respectively.
Mean terminal elimination half-life (t1/2) was3.50(±1.06) hours on day 14.
Mean trough plasma concentration of SUL-238 was 39.23(±24.31) ng/mL and 41.49(±18.20) ng/mL on day 8 and day 14, respectively.
Second cohort PK (1500 mg t.i.d.):
SUL-238 was rapidly absorbed, with a mean time to maximum plasma concentration (Tmax) reached at 0.95(±0.16) and 1.00(±0.00) hours on day 1 and day 14, respectively.
Mean terminal elimination half-life (t1/2):3.74(±1.84) hours on day 14.
Mean trough plasma concentration of SUL-238 was 57.98(±31.08) and 60.63(±64.14) ng/mL on day 8 and day 14, respectively.
Abidin Gülmüş, Chairman of GEN, stated:
"We're greatly motivated by these new positive results of SUL-238 in our Phase 1 trial, which mark a key advance toward addressing Alzheimer's disease at its biological foundation."
Nadir Ulu, MD, PhD, Vice President of R&D at GEN, added:
"With its excellent safety and PK profile in this Multiple Ascending Dose Phase 1 trial, SUL-238 continues to represent a very strong drug candidate for further clinical development aimed at meeting the critical unmet needs in neurodegenerative diseases, including Alzheimer's disease.
About SUL-238
SUL-238 is a novel, first-in-class, hibernation-derived small molecule designed to target mitochondria, the 'powerhouse' of the cell. SUL-238 supports mitochondrial bioenergetics via complex I/IV activation and enhances mitochondrial function in various preclinical models for neurodegenerative, cardiovascular, and renal diseases, as well as in accelerated aging. SUL-238 exhibits the capability to cross the blood-brain barrier and has undergone extensive safety evaluation in preclinical and clinical Phase 1 studies. GEN licenses SUL-238 from Sulfateq B.V. for neurodegenerative disease applications.
About GEN:
Founded in 1998, GEN is Türkiye's leading specialty pharmaceutical company, focused on developing innovative therapies across multiple therapeutic areas. Through significant R&D investments and global collaborations, GEN is committed to advancing healthcare worldwide. The company develops and manufactures high-quality, competitive products at its GMP-certified production facility and continues its bold efforts in original drug development via two dedicated R&D centers.
About Sulfateq:
Sulfateq B.V. is an early-stage Dutch biotech company that fosters strategic collaborations with academic and industrial research centres to accelerate the development of innovative new medicines. It has developed a novel class of small molecules, the SUL-compounds, that maintain mitochondrial health.
For more information:
www.genilac.com.tr
www.sulfateqbv.com
Contact Information
Bulutay Güneş
Sr. Head of Corporate Brand
[email protected]
Fatih Gören
Investor Relations Manager
[email protected]
SOURCE: GEN İlaç ve Sağlık Ürünleri A.Ş.
View the original press release on ACCESS Newswire
D.Johnson--AT

